lec09_2015S - Andrew.cmu.edu
advertisement
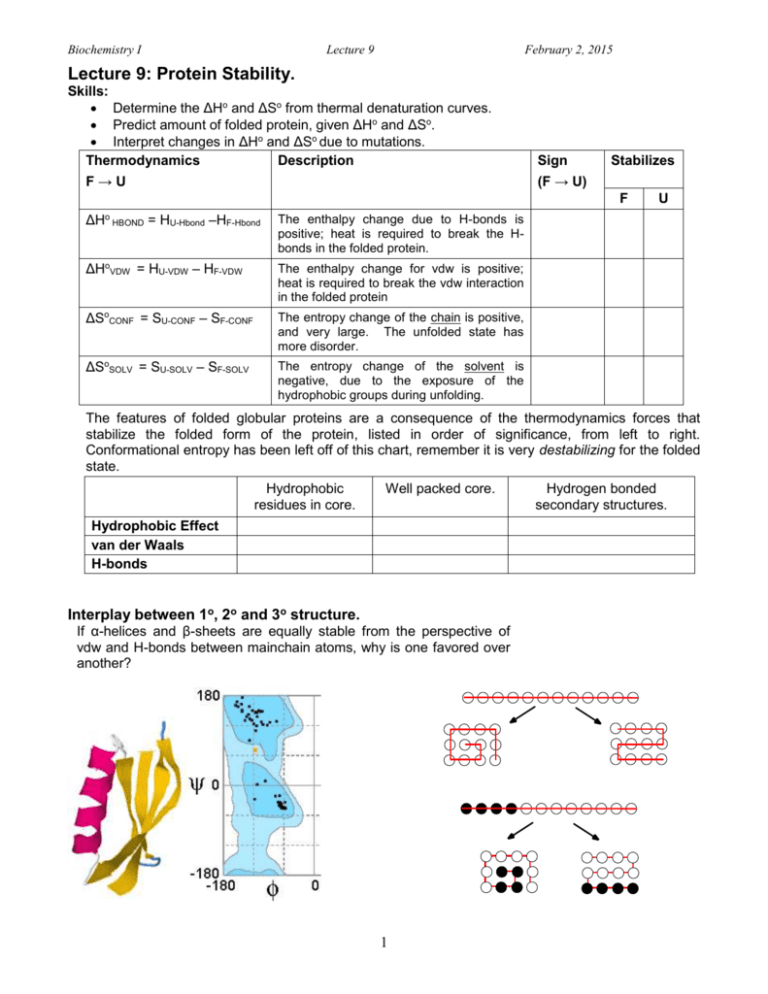
Biochemistry I Lecture 9 February 2, 2015 Lecture 9: Protein Stability. Skills: Determine the ΔHo and ΔSo from thermal denaturation curves. Predict amount of folded protein, given ΔHo and ΔSo. Interpret changes in ΔHo and ΔSo due to mutations. Thermodynamics Description F→U Sign Stabilizes (F → U) F ΔH o HBOND = HU-Hbond –HF-Hbond U The enthalpy change due to H-bonds is positive; heat is required to break the Hbonds in the folded protein. ΔHoVDW = HU-VDW – HF-VDW The enthalpy change for vdw is positive; heat is required to break the vdw interaction in the folded protein ΔSoCONF = SU-CONF – SF-CONF The entropy change of the chain is positive, and very large. The unfolded state has more disorder. ΔSoSOLV = SU-SOLV – SF-SOLV The entropy change of the solvent is negative, due to the exposure of the hydrophobic groups during unfolding. The features of folded globular proteins are a consequence of the thermodynamics forces that stabilize the folded form of the protein, listed in order of significance, from left to right. Conformational entropy has been left off of this chart, remember it is very destabilizing for the folded state. Hydrophobic residues in core. Well packed core. Hydrophobic Effect van der Waals H-bonds Interplay between 1o, 2o and 3o structure. If α-helices and β-sheets are equally stable from the perspective of vdw and H-bonds between mainchain atoms, why is one favored over another? 1 Hydrogen bonded secondary structures. Biochemistry I Lecture 9 February 2, 2015 Thermal Denaturation of Proteins: The relative energy of the native and unfolded state can be changed with temperature. Unfolding occurs at high temperature due to the positive ΔSo: G 0 H o TS o 1 0.9 Fraction Unfolded 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0 300 310 320 330 340 350 360 Tem p (K) Go = -RTlnKEQ Obtaining ΔHo and ΔSo: Predict fn, fu Exp Data Ho , So Calculate Go= Ho - T So Obtaining H0: The temperature dependence of the equilibrium constant can be used to determine Ho. Equating the two expressions for Go, and performing a little algebra: o o o This is the equation of a straight G H TS RT ln K EQ ln K EQ line, if lnKEQ is plotted versus 1/T. H o 1 S o R T R Slope = -Ho /R Ho = -slope R This plot is referred to as a van't Hoff Plot. Steps: 1. Vary temperature, record T & fraction native. 2. Convert temperature from C to K (C+273). 3. Calculate fraction unfolded, fU = (1-fN). 4. Calculate KEQ = fU/fN 5. Plot ln(Keq) versus 1/T (K). 6. Use the graphing wizard in Excel (Scatter plot). 7. Select plotted data with mouse. 8. Under Chart options, select "Add Trendline". 9. Use linear, under "Options" select "Display equation on chart". Obtaining S : Once the enthalpy is known, the change in entropy can be calculated from the TM and from Ho. At the melting temperature, TM, the energy difference between the native and unfolded states is zero. o 2 6 4 y = -24188x + 72.202 2 0 q e-2 K ln -4 -6 -8 -10 0.0027 0.0029 0.0031 1/T 0.0033 Biochemistry I Lecture 9 February 2, 2015 Example Question 1: Given Ho and So, predict the fraction [F ] [U ] unfolded at any given temperature. fF fU [ F ] [U ] [ F ] [U ] You work for a company that uses an enzyme to make the amino acid Lysine, an important amino acid in dinosaur food at Jurassic park. Your supervisor tells you to increase [ F ] /[ F ] [U]/[F] the production of lysine. The reaction is normally run at 290 [ F ] [U ] [F] [U ] K (~ room temperature). You know that the rate of the [F ] [F ] [F] [ F ] reaction, and therefore the Lysine production, will increase at K EQ 1 higher temperatures. Consequently, you increase the fF fU o 1 K EQ 1 K EQ reaction temperature to 310K (37 C). Given the following thermodynamic properties of the unfolding of the enzyme used in the reaction: Ho= +300 kJ/mol, So= +1000 J/mol-K, have you just lost your job? (RT=2.57 kJ/mol at 310 K). i. Calculate Go at the required temperature, Go=Ho-TSo Go = 300 – (310)x(1.0) = -10 kJ/mol ii. Calculateo KEQ at the required temperature: K EQ e G / RT = e+10/2.57 = e4 = 48.9 CH3 iii. Calculate fU using KEQ: fu = 48.9/(1+48.9) = 0.98. H Example Question2: The denaturation curves for both wild-type and mutants of a protein were measured to obtain Ho and So. Ho So Tm Wild-Type 210.0 kJ/mol 616 J/mol-deg 341 K N OH H N O Mutant 206 kJ/mol 611 J/mol-deg 337 K Threonine(Thr) S CH3 O Methionine(Met) 1.0 Wild-type Fu 0.5 Explain the effects on both H and S . o o Mutant 0 Denatured (Unfolded) Native (Folded) Wild-type O Thr 53 H=+210 kJ/mol CH3 O 337 341 Temperature O O O CH3 O O H H O Thr 44 H S=+616 O O CH3 O Altered Protein Met53 S CH3 O O H=+206 kJ/mol O S CH3 O O H S=+611 O O Thr44 O CH3 O SoH2O SoH2O SoH2O SoCONF (except 53) SoH2O (Thr53) So OBS = 616 3 SoH2O SoCONF (except 53)