Solubility
advertisement

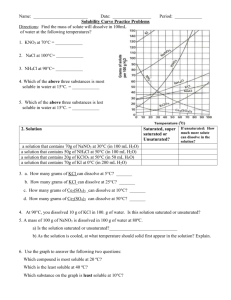
SOLUBILITY TAKS QUESTIONS July 2004 – 11: (23) The table shows temperature and pressure in four containers holding the same amount of water. According to the table, in which container can the most sodium chloride be dissolved in the water? AQ BR CS D* T Spring 2003 – 10: (50) Which of the following salts has the greatest solubility in water at 25°C? F CaCO3 G FeS H HgCl2 J* KClO4 Spring 2003 – 11: (10) A 0.2 g crystal of gypsum dissolves very slowly in 100 mL of water while the water is stirred. Which of these would cause the gypsum to dissolve faster? F Decreasing the water temperature G Stopping the stirring H Lowering the air pressure J* Crushing the crystal Spring 2003 – 11: (17) All of these can affect the rate at which a solid dissolves in water except — A* decreasing air pressure B stirring the water C increasing the temperature of the water D using larger crystals of the solid April 2004 – 10: (9) As a scuba diver goes deeper underwater, the diver must be aware that the increased pressure affects the human body by increasing the — A body’s temperature B* amount of dissolved gases in the body C amount of suspended solids in the body D concentration of minerals in the body Spring 2004 – 11: (39) Over time an open soft drink will lose carbonation (dissolved CO2). Which of these allows the CO2 to remain in solution the longest? A Reduced air pressure B Exposure to direct sunlight C Increased air currents D* Cooler temperatures February 2006 – 11: 50 MTBE is a gasoline additive that has entered some groundwater supplies. According to the table above, which of the following water temperatures would allow 79 grams of MTBE to dissolve in 1 liter of water? F* 0°C G 10°C H 30°C J 40°C Fall 2005 – 11, April 2006 - 11: 47 Nine groups of students dissolved as much potassium chloride as possible in water. Each group used 100 mL of water heated to a different temperature. Which graph shows the relationship between solubility and temperature for potassium chloride? Answer: D April 2006 – 11: 21 Power plants that discharge warm water into rivers have a negative effect on aquatic life. This is because the higher water temperature — A increases the pressure of the river water B increases the pH value of the river water C decreases sediment solubility in the river water D* decreases the dissolved oxygen in the river water April 2006 – 10: 37 A recipe calls for 210 grams of sugar to be dissolved in 0.25 liter of water. After the mixture is stirred, some sugar crystals remain in the water. What can be added to the mixture to help dissolve the remaining sugar crystals? A* Thermal energy B 2.0 g of baking soda C Ice cubes D 2.0 g of sodium chloride