Exam Review 1 - Iowa State University
advertisement

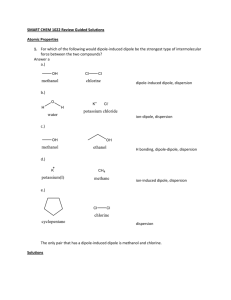
EXAM REVIEW 1 Supplemental Instruction Iowa State University Leader: Course: Instructor: Date: Prina Chem 178 Dr. Meyer 02/10/2015 1. Of the following options, which one best describes the relative strength of intermolecular interactions? a) (strongest) hydrogen bond > ion-dipole > dispersion (weakest) b) (strongest) dispersion > dipole-dipole > ion-dipole (weakest) c) (strongest) hydrogen bond > dispersion > ion-ion (weakest) d) (strongest) Ion-dipole > dipole-dipole > dispersion (weakest) 2. Indicate the primary interaction observed in the following solutions: a) KCl in water b) CH2Cl2 in C6H6 c) CH3OH in water 3. When ammonium chloride dissolves in water, the solution becomes colder. The solution process is: a) endothermic b) exothermic c) no heat exchange takes place. 4. The solubility of Cr(NO3)3•9H2O in water is 208 g per 100 g of water at 15 ºC. A solution of Cr(NO3)3•9H2O in water at 35 ºC is formed by dissolving 324 g in 100 g water. When this solution is slowly cooled to 15 ºC, no precipitate forms. What term describes this solution? A) immiscible B) unsaturated C) supersaturated D) crystallized E) saturated 5. What is the osmotic pressure (in atm) of a 0.040 M solution of a non-electrolyte at 30.0 C? a. 2atm b. 5atm c. 1 atm d. 2.5atm 6. Which one of the following substances would be the most soluble in CCl4? a. CH3CH2OH b. H2O c. NH3 d. C10H22 e. NaCl 7. The solubility of nitrogen gas at 25°C and 1 atm is 6.8 x 10-4 mol/L. If the partial pressure of nitrogen gas in air is 0.76 atm, what is the concentration (molarity) of dissolved nitrogen? a. 6.8 × 10-4 M b. 5.2 × 10-4 M c. 4.9 × 10-4 M d. 3.8 × 10-4 M e. 1.1 × 10-5 M 8. The concentration of nitrate ion in a solution that contains 0.900 M aluminum nitrate is __________ M. a. 0.900 b. 0.450 c. 0.300 d. 2.70 e. 1.80 9. The concentration of lead nitrate (Pb(NO3)2) in a 0.726 M solution is __________ molal. The density of the solution is 1.202 g/mL. a. 0.476 b. 1.928 c. 0.755 d. 0.819 e. 0.650 10. A solution is prepared by dissolving 15.0 g of NH3 in 250.0 g of water. The density of the resulting solution is 0.974 g/mL. Find the resulting molarity of the solution. ___M a. 0.640 b. 0.0597 c. 0.940 d. 3.24 e. 16.8 11. Molality is defined as the __________. a. moles solute/moles solvent b. moles solute/Liters solution d. moles solute/kg solvent e. none (dimensionless) c. moles solute/kg solution 12. What is the molecular weight of a nonelectrolyte if 5.0 g dissolved in 100.0 g of water has a freezing point of -0.25°C? Kf=1.86 C/m a. 370g/mol b. 190g/mol c.37g/mol d.20g/mol 13. What is the freezing point of 0.50 m NaCl(aq)? Kf=1.86 C/m a. 0.93 C b.-0.93C c.0.52C d.-1.9C 14. Which of the following when dissolved will create the highest vapor pressure? a. 0.15m propane b. 0.2m KBr c. 0.1mMgCl2 d. 0.2m methanol 1060 Hixson-Lied Student Success Center 515-294-6624 sistaff@iastate.edu http://www.si.iastate.edu 15. Consider the following reaction CO2 (g) + H2 (g) <-> CO (g) + H2O (g) Calculate the value of the equilibrium constant, Kc , for the above system, if 0.1908 moles of CO2, 0.0908 moles of H2, 0.0092 moles of CO, and 0.0092 moles of H2O vapour were present in a 2.00 L reaction vessel at equilibrium. 16. Consider the following reaction. The initial concentrations are [HSO4–] = 0.50 M, [H3O+] = 0.020 M, [SO42– ]= 0.060 M. HSO4– (aq) + H2O (l)<-> H3O+ (aq) + SO42– (aq) Kc = 0.012 (a) Which way would the reaction shift to reach equilibrium? (b) What are the equilibrium concentrations of the products and reactants. 17. Consider the decomposition of nitrous oxide, laughing gas, 2N2O (g) <->2 N2 (g) + O2 (g) At 25 C, Kc is 7.3 x 10^34. (a) Based on the information given, which side does the reaction favor? (b) What is the value of Kp for the reaction at 25 C? 18. Ammonia, NH3, is produced by the following reaction: N2(g) + 3H2(g) 2NH3(g) + energy Which of the following would result in the highest concentration of ammonia at equilibrium? A. increasing the temperature and increasing the pressure B. decreasing the temperature and increasing the pressure C. increasing the temperature and decreasing the pressure D. decreasing the temperature and decreasing the pressure 19. Consider the following equilibrium : SO2Cl2(g) + energy ⇄ SO2(g) + Cl2(g) When the temperature is decreased, the equilibrium shifts A. left and the [SO2Cl2] increases B. left and the [SO2Cl2] decreases C. right and the [SO2Cl2] increases D. right and the [SO2Cl2] decreases 20. When hydrogen iodide (HI) is placed in a closed container it will decompose into its elements hydrogen and iodine and an equilibrium will be established: 2 HI (g) <-> H2(g) + I2(g) If the initial concentration of HI and the equilibrium constant for the system are known, then we can calculate the equilibrium concentration for HI, H2, and I2. Initial concentration of HI: 0.025 mol/L K c=2.2x10-3 21. If you dissolved 28 grams of NaCl in 90 grams of pure H20 hot enough to have a vapor pressure of 30 torr, what will the new vapor pressure be? 22. If 152 g of sodium sulfate, Na2SO4, are dissolved in 875 g of H2O, what will be the boiling point of the resulting solution? Assume 100% ionization. Kf=1.86 C/m GOOD LUCK!