redox
advertisement

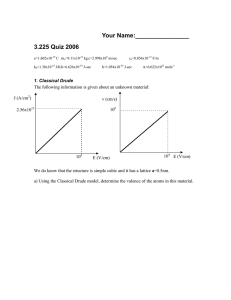
Name: _________________________________________ Date:____________________________ Optional Homework 6 – for practice only!!! 1.) Write the oxidation number for each atom in the molecules below: a. Gallium sulfide b. Chromium (III) oxide c. Potassium phospite d. Calcium phosphate e. Aluminum carbonate 2.) Write the half-reactions for each of the following chemical reaction including the electrons and indicate exactly which species is oxidized, which is reduced, which is the oxidizing agent and which is the reducing agent: they are not balanced – please do no worry/balance them!! a. PbO (s) + CO (g) → Pb (s) + CO2 (g) b. Fe(s) + Cl2 (g) → FeCl3 (s) c. CO (g) + I2O5 (s) → I2 (s) + CO2 (g) d. Cu (s) + HNO3 (aq) → Cu(NO3)2 (aq) + NO2 (g) + H2O (l) 1.) Balance the following chemical reactions as indicated identify the reducing and oxidizing species: (insert spaces as needed or perform on a separate sheet of paper) a. SO3-2 (aq) + Cl2 (g) SO4-2 (aq) + Cl-1(aq) [basic] 2.) Balance the following chemical reactions as indicated identify the reducing and oxidizing species: (insert spaces as needed or perform on a separate sheet of paper) a. MnO4-1 (aq) + H2O2 (aq) Mn+2 (aq) + O2 (g) [acidic] 3.) Consider the following general voltaic cell: voltmeter A B E C D Identify the anode, cathode, salt bridge, electrode at which the electrons accumulate (negatively charged electrode), the electrode at which the electrons are consumed (the positively charged electrode), the electrode that would gain mass as the cell operates (assuming that the metal plates out of solution), and the electrode that would lose mass as the cell operates 4.) Using the activity series, predict which of the following reactions will occur spontaneously AS WRITTEN: remember that with single replacement reactions you can form H2 gas a. Cu(s) + Zn(NO3)2(aq) b. Al(s) + Pb(NO3)2(aq) c. Al(s) + HCl(aq) d. Mg(s) + HCl(aq) e. Fe(s) + CuSO4(aq) f. Fe(s) + AgC2H3O2(aq) g. Fe(s) + NaBr(aq) h. AgNO3(aq) + Cu(s) 5.) CALCULATE Eo for the oxidation half-reaction Co(s) Co+2(aq) + 2e- given the cell voltage for the following voltaic cell (Platinum does not participate in the overall chemical reaction) Co(s) Co+2 (1 M) Ce+4 (1 M), Ce+3 (1 M) Pt(s) Eocell = 1.887 V Ereduction = 1.44 V 6.) Balance the following given half-reactions and determine the overall balanced redox reaction and the Eocell for the overall reaction: O2 (g) H2O (l) and I-1 (aq) → I2 (s) 7.) Determine Eocell values for the following redox reaction: 2Al(s) + 3Cu+2(aq) Al+3(aq) + 3Cu(s) 8.) Will copper metal displace silver ion from aqueous solution? That is, does the reaction occur spontaneously from left to right or right to left. Prove this mathematically by calculating Eocell. Cu(s) + 2Ag+1(aq) Cu+2 + 2Ag(s) 9.) Write the electron configurations – DO NOT USE THE NOBLE GAS CONFIGURATION!! – for the following: a. Fe b. Zn 10.) Write a set of quantum numbers (n, l, ml, and ms) for the 5th electron, the 10th electron, the 12th electron, the 20th electron, the 27th electron, and the 30th electron for zinc. (HINT!! Orbital box diagrams might help!) 11.)Write the electron configurations – DO NOT USE THE NOBLE GAS CONFIGURATION!! – for the following a. K+1 b. Cr+3 c. Cr+6 12.) Which of the following quantum number sets are allowed? If there is an error identify AND correct it by changing the l number only a. n= 3, l = 2, ml = 2 b. n=6, l =5, ml = -4