Chapter 9 - Fayetteville State University
advertisement

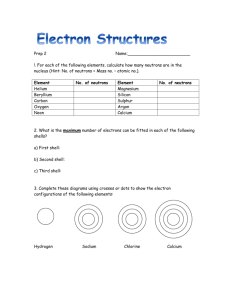
-1FAYETTEVILLE STATE UNIVERSITY COLLEGE OF BASIC AND APPLIED SCIENCES DEPARTMENT OF NATURAL SCIENCES CSCI 110-04 FALL 2005 Nov. 21-05 Chapter 9 The Periodic Law I) Basic concepts and Equations 1) Elements: Are the simplest substances from which matter is made up, they can not be decomposed into new elements by chemical means. 2) Compounds: Substances made up of elements which are combined in very well defined proportions, compounds can be decomposed in their components by chemical means. 3) Mixtures: Are composed of elements of compounds, where the elements or compounds in the mixture retain their unique properties. 4) Solutions: are homogenous mixtures of elements of compounds 5) Atoms: are the ultimate particles of elements 6) Molecules: are made of atoms and form the ultimate particles of gaseous or liquid compounds 7) Periodic Law: States that elements arranged in order of the atomic number share similar chemical and physical properties. These arrangement are called groups, examples are the alkali metals (Li, Na, K, Rb, Cs), the halogens (F, Cl, Br, I). 8) Groups: A sequence of elements of increasing atomic number in the periodic table that share similar chemical properties (Example: group 1A Li, Na, K, Rb, Cs) 9) Metals: are characterized for having extra electrons outside the closed shells, these electrons are easily lost when metals combine with nonmetals. 10) Nonmetals: These atoms have open shells of one of more electrons, which allow them to form compounds with metals by picking up readily available electrons of the latter atoms. 11) Covalent Bond: is defined as the interaction between atoms where a pair of electrons is shared. -212) Ionic Bond: The interaction between the atoms is due to transference of electrons from atom to the other, which cause a strong interaction between the ions formed. 13) Atoms Groups: atoms can interact between them to form stable positive of negative ions, examples are the sulfate and the nitrate ion (SO4= and NO3-). 14) Chemical equation: can be defined as the equality between chemical species to describe a chemical reaction. In these expressions the number of atoms of each kind are equal a both sides of the equation, example: 2H2 + O2 2H2O 4H 4H 2O 2O II Tests 1. A pure substance that cannot be decomposed by chemical means is a. an element b. a compound c. a solid d. a solution 2. Elements can be distinguished unambiguously by their a. hardnesses b. colors c. atomic numbers d. electrical properties 3. The number of known elements is approximately a. b. c. d. 50 100 200 500 4. At room temperature and atmospheric pressure, most elements are a. gases b. liquids c. metallic solids d. nonmetallic solids 5. Which of the following substances is a homogeneous mixture? a. iron b. seawater c. salt d. paper 6. Which of the following substances is a compound? a. iron b. seawater c. salt d. paper 7. The nonmetal whose chemical behavior is most like that of typical metals is a. hydrogen -3b. helium c. chlorine d. carbon 8. Which one or more of the following properties are characteristic of all metals? a. Conducts electricity well b. Conducts heat well c. Is a solid at room temperature d. Is transparent to light 9. Iodine is an example of a. an inert gas b. an alkali metal c. a halogen d. a compound 10. Of the following metals, the most active chemically is a. gold b. aluminum c. iron d. sodium 11. Of the following metals, the least active chemically is a. gold b. calcium c. lead d. sodium 12. Of the following nonmetals, the most active chemically is a. b. c. d. helium fluorine oxygen sulfur 13. At room temperature, chlorine is a. a colorless gas b. a greenish-yellow gas c. a reddish-brown liquid d. a steel-gray solid 14. Of the following nonmetals, the one not a halogen is a. fluorine b. bromine c. sulfur d. iodine 15. The place of an element in the periodic table is determined by its a. atomic number b. atomic mass c. density d. chemical activity 16. Each vertical column of the periodic table includes elements with chemical characteristics that are, in general, a. identical b. similar -4c. different d. sometimes similar and sometimes different 17. The periodic table of the elements does not a. permit us to make accurate guesses of the properties of undiscovered elements b. reveal regularities in the occurrence of elements with similar properties c. include the inert gases d. tell us the arrangement of the atoms in a molecule 18. The elements in group 1 of the periodic table (except for hydrogen) are a. all metals b. all nonmetals c. both metals and nonmetals d. neither metals nor nonmetals 19. Of the elements in group 8 of the periodic table, at room temperature and atmospheric pressure a. all are gases b. all are liquids c. some are gases and the others liquids d. some are liquids and the others solids 20. An alkali metal atom a. has one electron in its outer shell b. has two electrons in its outer shell c. has a filled outer shell d. lacks one electron of having a filled outer shell 21. A halogen atom a. has one electron in its outer shell b. has two electrons in its outer shell c. has a filled outer shell d. lacks one electron of having a filled outer shell 22. An inert gas atom a. has one electron in its outer shell b. has two electrons in its outer shell c. has a filled outer shell d. lacks one electron of having a filled outer shell 23. The most important factor in determining the chemical behavior of an atom is its a. nuclear structure b. electron structure c. atomic mass d. solubility 24. An atom that loses its outer electron or electrons readily is a. an active metal b. an active nonmetal c. an inactive metal d. an inactive nonmetal 25. When they combine chemically with metal atoms, nonmetal atoms tend to a. gain electrons to become negative ions b. lose electrons to become positive ions c. remain electrically neutral -5d. any of these, depending upon the circumstances 26. When atoms join to form a molecule, a. energy is absorbed b. energy is given off c. energy is neither absorbed nor given off d. any of these, depending on the circumstances 27. In a covalent molecule, a. at least one metal atom is always present b. one or more electrons are transferred from one atom to another c. adjacent atoms share one or more electrons d. adjacent atoms share one or more pairs of electrons 28. An element that can form an ionic compound with chlorine is a. carbon b. copper c. sulfur d. neon