Chapter 10 - Geology and the Gem Minerals
advertisement

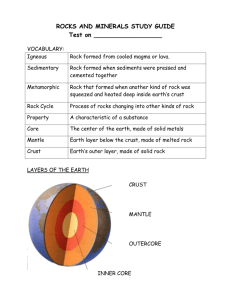
CHAPTER 10 – GEOLOGY AND THE GEM MINERALS This chapter is a simple outline of those geological environments which favor the formation of gem minerals. The earth is a dynamic planetary body continuously changing through the activities of two cyclic phenomena. The weather cycle is driven by energy from the sun. It involves circulation of water within the earth’s atmosphere and hydrosphere and alters rocks at the earth’s surface by the process of weathering. Sedimentary rocks are formed by the weather cycle. Near surface weathering of feldspar and other primary minerals yields clay with a byproduct of colloidal silica which is the source of opal, agate and the chalcedony varieties. The tectonic cycle is driven by energy from the earth’s interior. It is a sluggish circulation, over geologic time, within the earth’ mantle which brings heat and light elements to the earth’s surface. Igneous and metamorphic rocks are formed by the earth’s tectonic cycle and are the host rock for the formation of most gem minerals. Structure of the Earth Like a hard-boiled egg, the earth has a yolk (core), a white (mantle) and a shell (crust) (Figure 10-1). The core of the earth is thought to be largely metallic iron and nickel. The outer core seems to have the low rigidity characteristic of molten metal, and currents within it may be the source of the earth’s magnetic field. The mantle of the earth surrounds the core like the white of the egg and represents about 80% of the earth’s volume. It appears to be very heavy rock, rich in magnesium and iron and relatively poor in silicon, aluminum and the alkali elements. The major mineral of this rock is 1 probably olivine (peridot) like that found in stony meteorites and the mantle xenoliths in basalt flows. Rock within the mantle is not totally rigid but moves slowly by plastic flow, thereby circulating heat from the core to the crust like the convection currents in boiling water. The crust of the earth, like the shell of the egg, is thin and rigid and may be broken into rigid plates which slip about on the earth’s surface in response to the circulating mantle rock beneath them. Eight large plates and numerous smaller ones make up the earth’s crust (Figure 10-2). Some plates are entirely ocean crust and other plates have floating masses of low-density continental rock which ride high on the ocean crust. Plate boundaries may divide ocean basins or split continents but tend to parallel continental margins. The ocean crust is thin, heavy, mafic and geologically young. It is composed almost entirely of basalt which is a dark, fine-grained volcanic rock, extruded largely in the ocean basins where crustal plates separate. Mafic (Mg-Fe) rocks are relatively silica poor (about 50% SiO2) and tend to be rich in magnesium, iron and calcium. Continental crust is thick, light weight, felsic and may be youthful to very old. Felsic (feldspar-silica) rocks are silica rich (about 70% SiO2) and tend to be rich in aluminum and the alkali elements sodium and potassium. Continental crust forms where crustal plates converge and is represented by a vast variety of rock types, which, summed together, have the composition of granite. The earth’s crust rests in isostatic equilibrium (Gr. isos = equal, Gr. statos = standing) on the underlying plastic mantle (Figure 10-3). Where weight is placed on the earth’s crust, it subsides, and where weight is removed, the crust rises. Where mountains stand elevated, the continental crust is thick, and light-weight crustal rock is pressed down into the plastic rocks of the mantle, like an iceberg floating in the sea. As the mountains wear down, their light-weight 2 roots float upward causing the mountains to rise, and the processes repeats until the mountains are eroded completely away, exposing the igneous and metamorphic core of the mountains (basement rock), and the roots of the mountains are raised to the level of the continental base. Plate Tectonics: The shell fragments of our egg paradigm can interface in only three ways. 1- They may pull apart, exposing the white of the egg. 2- One fragment may slip underneath another fragment. 3- Fragments, with relatively straight edges, may slip past one another without separation or overlap. If we were to trace the outer edge of any specific shell fragment, we would note that everywhere the edge is one of the three types, i.e., one might see a “pull apart” edge on one side of the fragment and a “slip underneath” edge on another side. Transform plate boundaries form where plates of the earth’s crust slip past one another, offsetting topographic features, without separation or overlap and are represented by the San Andreas Fault, in southern California, or the North Anatolian Fault, in northern Turkey. These large strike-slip faults are characterized by shallow earthquakes but no important gem-making processes. Spreading centers form where the earth’s crustal plates pull apart (Figure 10-4). Plate separation releases pressure on the underlying mantle rock which partially melts* to form the hot, fluid lavas of the basalt flows which make up the ocean floor. Ocean crust is Created at spreading centers and, where a spreading center divides a continental mass (e.g., East African rift valley), a long narrow sea (e.g., Red Sea) heralds the beginning of a broad, basaltic ocean basin (e.g., Atlantic Ocean) Characteristic of spreading centers are: shallow earthquakes, 3 mild fissure-eruption flood basalts (Figure 10-13), and rift valleys (e.g., Jordan Valley) flanked by highlands (e.g., Judean Wilderness and Jordanian Highlands), which are the result of thermal expansion of the crust due to heat escape from the mantle. The only gem mineral directly associated with the spreading centers is peridot, which may be brought up from the mantle rock as phenocrysts (Gr. phaino = apparent, Gr. krystallos = crystal) in basalt (Figure 10-15B). *Partial melting implies that some minerals of the rock will melt while others do not. Every crystalline mineral has a specific melting point, and the more silica rich the mineral the lower its melting temperature. Therefore, the silica rich minerals melt first, yielding a molten phase (magma) which is more felsic than the original rock and a solid residue that is more mafic. By this process, felsic elements (Si, Al, Na, K) are brought to the earth’s surface as continental crust, and the mafic elements (Mg, Fe, Ca) descend into the ultramafic rocks of the earth’s mantle. Subduction zones form where one crustal plate under-rides another (Figure 10-5). The under-riding plate is always ocean crust, but the over-riding rock mass may be either ocean crust or continental crust. The under-riding plate plunges into the mantle where it is partially absorbed into the mantle rock and partially melts to form the low-temperature, viscous magma of granitic batholiths (Gr. bathos = deep, Gr. lithos = stone) and violent volcanoes. Converging crustal plates generate strong compressional forces. If the subduction zone lies along a continental boundary, these compressional forces fold the off-shore sediments forming a mountain belt of sedimentary rock parallel to the continental margin (e.g., Andes Mts. of South America). In the deep core of the mountain system, these sedimentary rocks are subjected to the high temperature and pressure of deep burial (i.e., metamorphism) and to the stress forces of compression and the temperatures of magmas generated by the partial melting of the subducting ocean crust. These felsic, viscous magmas tend to rise through the 4 metamorphosed sediments, due to the lower specific gravity of the magma, and may burst through in the tops of the overlying mountains as violent stratovolcanoes (e.g., volcanoes of the Cascade Range of Washington and Oregon) (Figure 10-14). More often, the viscous, felsic magmas cool at depth beneath the mountain range as large granite batholiths, surrounded by high grade metamorphosed sediments. This complex of granite batholiths and high grade metamorphic rocks, commonly called “basement complex”, is exposed at the surface by the uplift and erosion described above, and herein we find many of the minerals we call gems. Chemistry of the Earth’s Crust: Ninety-two elements occur naturally in the rocks of the earth’s crust, however, only eight occur in amounts greater than one weight percent (see Table 1-2). All common minerals are some combination of these eight elements and all other elements are considered trace elements. Since oxygen makes up half of the earth’s crust, by weight, most ionic mineral structures are an arrangement of large oxygen anions with small, interstitial cations holding the oxygen anions together to form the crystal structure. And, since silicon makes up half of the remaining 50%, most minerals are silicates. The most common minerals in the earth’s crust are shown in Table 10-1. We are familiar with quartz as sand grains or pebbles, but the other common minerals are less familiar to us, because they are not stable at the earth’s surface where they are exposed to the water and gasses of the earth’s atmosphere (i.e., weathering). Feldspars and the ferromagnesian minerals (pyroxenes and amphiboles) decompose to clay or mud, with byproducts which enrich the salinity of the oceans and cement the sand and pebbles to produce sandstones, conglomerates and other detrital sedimentary rocks. Ferric hydroxide and colloidal 5 silica byproducts color the sediments with rust and form veins of chalcedony and opal in the thin blanket of sedimentary rock which covers 80% of the land mass. Besides the colloidal silicas (e.g., opal and agate), a few phosphates (e.g., turquoise and variscite) and carbonates (e.g., malachite and calcite) form in this sedimentary blanket and qualify as gem minerals. The great majority of familiar gems are formed in the granites and metamorphic rocks of the underlying “basement”, exposed in the heart of modern mountain systems or on the low rolling hills or plains of continental shields, where the deeply eroded roots of ancient mountains are exposed (Figure 10-5). Table 10-1 Mineral Abundance in the Earth’s Crust Feldspar Mineral Group 60% Alkali Feldspar (20%) Plagioclase Feldspar (40%) Amphibole and Pyroxene 17% Quartz 12% Micas 4% Olivine 1% Magnetite and Ilmenite 1% 95% The above figures represent the approximate mineral abundance in the earth’s crust but do not represent the mineral abundance at the earth surface. In the above mineral list, only quartz 6 is truly stable at the earth’s surface. Olivine, pyroxene, amphibole and feldspar break down on exposure to the earth’s atmosphere to yield various varieties of clay. The micas and magnetite, although moderately stable, will decompose with time to clay and rust. Igneous Rocks - Plutonic (Intrusive) Granite and Granite-like Rocks (Figure 10-6) - Most intrusive igneous rocks are granite formed as a huge, shapeless mass (batholith) of cooling magma at depths of several kilometers in subduction zones (Figure 10-5). A granite magma is a chemical system of elements resulting from the melting of select minerals (i.e., those minerals with the lowest melting points) in the ocean crust being subducted. Granite rock is coarse-grained, because the magma cools slowly at depth, and consists largely of feldspar varieties (K-feldspar and plagioclase) and quartz. The granite-like rocks (e.g., granodiorite, quartz monzonite, syenite, etc.) differ only in the relative proportions of quartz and the feldspar varieties. A granite magma is a chemical system at a temperature above the melting point of its minerals. Each mineral crystallizes from the system when the magma cools to its crystallization temperature (i.e., melting point temperature), and each mineral takes from the magma those elements required by its composition. The earliest (i.e., highest temperature) minerals to crystallize are as mafic as the magma composition can allow and are more mafic than the magma they crystallized from. From a granitic magma, the first major minerals to form might be an amphibole [e.g., hornblende Ca2(Mg,Fe+2)4(Al,Fe+3)(Si7Al)O22(OH)2] or dark mica, e.g., biotite [K2(Mg,Fe+2)5Al(Si5Al3)O20(OH)2], which will probably be the only dark (i.e., mafic) mineral in the granite. Contemporaneous with the dark minerals will be a Ca-rich feldspar (plagioclase) which will become more Na-rich as the crystallization proceeds. The final minerals in the main 7 crystallization sequence of a granitic magma will be orthoclase feldspar [K(Si3Al)O8] and quartz [SiO2], yielding a solid rock of quartz, two feldspars (K-rich orthoclase and Na-rich plagioclase) and minor dark minerals. Quartz is the left-over silica, after the other major elements are exhausted from the magma, and it crystallizes last (i.e., lowest temperature) or melts first from a granite rock. Dark “granites”, like gabbro or diorite (Figure 10-7), are much less common than the light granites described above. They are coarse-grained, like granite, but contain major percentages of iron-magnesium-rich silicates (mafic) which provide the overall dark color. They may contain neither quartz nor alkali-feldspar (i.e., K-rich feldspar or Na-rich feldspar) and consist largely of Ca-rich plagioclase feldspar [Ca(Si2Al2)O8] and pyroxene [Ca(Mg,Fe+2)(SiO3)2], sometimes with olivine [(Mg, Fe+2)2SiO4]. For the more mafic minerals to form, we would have to begin with a magma more rich in iron, magnesium and calcium than the common granitic magmas, and such magmas are less common in subduction zones. Pegmatites and Hydrothermal Veins - Although pegmatites (Figure 10-8) and hydrothermal veins are almost an afterthought in the crystallization of granite magma, we consider them here because they are the host for a great variety of gem minerals. Only the big eight elements (O, Si, Al, Fe, Ca, Mg, Na, K) are likely to be represented in the major minerals of a common granite. A typical granitic magma may, however, contain five or more weight-percent of dissolved water, only a small percentage of which may be forced into the amphiboles and micas as the hydroxide anion (OH)−. The final magma phase is very silica-rich and very volatile-rich (i.e., H2O, CO2, SO2, H2S, etc.). Steam and carbon-dioxide are odorless, but the sulfur gasses we expect to smell in thermal spring areas, where they escape from cooling 8 magmas below (Figure 10-9). The final magma fluids are relatively low temperature but highly fluid, due to their high volatile content, and they contain all the trace elements, incompatible in the structures of the common minerals of a granite. Burial pressure on the underground magma chamber may squeeze the interstitial fluids from the cooling granite body, forcing them into the enclosing country rock as distinct pegmatite dikes or hydrothermal veins (Figure 10-8), or these volatile-rich fluids may merely saturate the surrounding rock along mineral grain boundaries (i.e., contact metamorphism Figure 10-25). A granite pegmatite dike is a potential jewel box made up largely of coarse crystals of the final minerals to crystallize from a normal granite magma, i.e., alkali feldspar [(K,Na)(Si3Al)O8], white mica [KAl2(Si3Al)O10(OH)2] and quartz [SiO2]. The mineral crystals may be very large, due to the high volatile (gas) content, which makes this residual magma highly fluid. Injected into pre-existing fractures in the enclosing rocks, this highly fluid magma is frozen against the cold wall rock as a fine-grained border zone of quartz, feldspar and mica. The intermediate and core zones cool more slowly and may form very large crystals in the presence of an aqueous fluid which separates from the remaining silicate magma. The intermediate zone may contain intergrowths of K-feldspar and quartz, in the proportion 72% K-feldspar and 28% quartz, which resembles cuneiform writing, hence the name “graphic granite”(Figure 10-10). Volatiles in the fluid may force open cavities in the core zones which are the “jewel boxes” of the pegmatite. The vast majority of pegmatites are simple pegmatites containing little more than the three basic pegmatite minerals, and most pegmatites form at great depths, associated with highgrade metamorphic rocks, where pressures are too high to allow volatiles to expand to form voids. Gem pegmatites are extremely rare and are complex pegmatites which contain the trace 9 elements essential to gem formation and which form at relatively shallow depths where highvolatile fluids separate from final-stage magmas to form “pockets” where gem minerals may grow, relatively unobstructed, into an open cavity. The most common trace elements in this phase are the light metals of the first period of the periodic table (i.e., lithium, beryllium and boron) and the volatile fluorine. Spodumene [LiAl(SiO3)2], beryl [Be3Al2(SiO3)6], tourmaline [Na(Li,Al)3Al6(SiO3)6(BO3)3(OH,F)4] and topaz [Al2(SiO4)(F,OH)2] are the most common resulting gem minerals, although many other pegmatite gem minerals are possible, e.g., chrysoberyl [BeAl2O4], phenakite [Be2SiO4], euclase [BeAlSiO4(OH)], amblygonite [LiAl(PO4)F], beryllonite [NaBePO4], petalite [Li(Si4Al)O10], etc.. We may expect pegmatite gem minerals to contain light trace elements in some combination with the felsic elements Si, Al, Na, K and we might anticipate crystals that could be faceted to yield quite large gemstones. A hydrothermal vein (Gr. hydro = water and Gr. therme = heat) forms from the aqueous solutions which remain after the last magma has crystallized and may grade away from a pegmatite dike when the alkali feldspar and white mica (muscovite) have mostly crystallized and little remains but silica and hot water, not greatly above its surface boiling point. The silica may crystallize as massive quartz, milky with tiny liquid inclusions, or as cryptocrystalline (Gr. krypto = hidden) quartz (i.e., chalcedony). Fluorine gas asserts itself in fluorite [CaF2] and cryolite [Na3AlF6] and sulfur gases may deposit sulfur crystals at fumerole vents (Figure 10-9) or form sulfates like gypsum [CaSO4·2H2O] or barite [BaSO4]. Milky quartz is a common host for native gold (Figure 7-6d), and a great variety of heavy metallic elements are concentrated in the final solutions and combine with sulfur to form sulfide ore deposits of mercury, arsenic, antimony, tin, tungsten, molybdenum, and the common base metals copper, lead and zinc. 10 Igneous Rocks - Volcanic (Extrusive) Vulcanism is a continuous spectrum from gentle basalt flows that flood the landscape to the violent explosions that blast away mountains, and most existing volcanoes fall near the ends of the spectrum. Mild volcanoes (Figure 10-11) are characteristic of plate spreading where basaltic lavas ooze onto the ocean floor through opening fissures, making vast beds of “pillow” basalt (Figure 10-12), as new ocean crust (e.g., Mid-Atlantic Ridge). Similar vulcanism results over mantle “hot spots” (e.g., Hawaiian Islands) where basaltic lavas originate in the upper mantle and rise through the crust to form island chains of giant shield volcanoes, as the ocean plate moves over the stationary hot spot in the mantle. Where crustal spreading divides a continent, fields of fissure eruption basalts (Figure 10-13) flood vast continental areas (e.g., Columbia River Plateau). Mild vulcanism is characterized by: Hot and fluid basaltic lavas Nearly 100% lava flows (i.e., little solid ash) Fissure eruption Gentle slopes of broad shield volcanoes Almost continuous eruption Molten lava in the throat of the volcano and lava lakes in the crater. Basalt - Basaltic magmas represent the high-temperature melting of iron-magnesium rich (mafic) silicates, usually where mantle rock is exposed at spreading centers. At spreading centers, highly fluid mafic magmas usually reach the surface as lavas which crystallize as a dark, fine-grained rock of olivine [(Mg, Fe+2)2SiO4], pyroxene [Ca(Mg,Fe+2)(SiO3)2], and Ca-rich 11 plagioclase [Ca(Si2Al2)O8], i.e., basalt (Figure 10-15). Olivine crystallizes first and may grow to significant size before being surrounded by fine-grained plagioclase (labradorite) and pyroxene in the final rock. Olivine (gem name “peridot”) in basalt is usually too small to be of practical value and forms the green sands of Hawaiian beaches. The violent volcanoes (Figure 10-14) are the consequence of plate subduction. They are the islands of oceanic island arcs (e.g., South Pacific islands) or appear in the youthful and growing mountain ranges along continental margins, inland of deep ocean troughs (e.g., Andes). Their violence may exceed the energy of many atomic bombs and some have altered the course of history and world climate (e.g., Vesuvius, Santorini, Krakatoa). Violent vulcanism is characterized by: “Cool” and highly viscous rhyolitic lavas Nearly 100% pyroclastics and ash flows Central eruption from giant, symmetrical volcanic cones Steep slopes of layered stratovolcanoes Intermittent eruption with long periods between eruptions Solid lava in the throat of the volcano, requiring huge pressure build up for eruption Andesite (Figure 10-16) is basalt, with more sodium-rich plagioclase (andesine) and no olivine. It is more felsic and less dark than basalt and commonly green or purple with visible plagioclase phenocrysts (Gr. pheno = visible and Gr. krystallos = crystal). Andesite is characteristic of huge stratovolcanoes like those in the Andes Mountains, from which it derives its name, and is associated with crustal plate subduction Rhyolite (Figure 10-17) is the volcanic equivalent of granite with the same chemical and 12 mineral composition. Rapid cooling at the earth’s surface, however, produces a light-colored, fine-grained volcanic rock, usually pink or pale lavender, where no individual crystals are visible, without magnification. Very rapid cooling of the viscous, rhyolitic lavas may yield uncrystallized volcanic glass, i.e., obsidian (Figure 10-18), which is usually black, owing to very tiny magnetite (Fe0.Fe2O3) crystals scattered throughout the transparent glass. As with granite, the final stages in the cooling of rhyolitic lava may yield highly mobile, low-temperature, pegmatite solutions of water and gasses rich in silica. These mineralizing solutions and gasses may force fractures and voids within the rhyolite or associated ash beds and, if endowed with unique pegmatite trace elements, they may form crystals, in fissures and cavities, of topaz, beryl or other minerals characteristic of pegmatites. Sedimentary rocks Sedimentary rocks (Figure 10-19) are stratified, secondary rocks, made from the physical break down or chemical decay of any pre-existing rock. Although igneous rocks make up most of the earth’s crust, rocks at the earth’s surface are largely sedimentary and form a thin blanket over the igneous-metamorphic basement. Although feldspar is by far the most abundant mineral group in the earth’s crust, it is largely absent in the sedimentary blanket, because feldspars are chemically unstable under surface conditions. Feldspars react with oxygen, water and carbondioxide in the air to decay to clays (i.e., mud) with byproducts of colloidal silica and soluble salts. 2Na(AlSi3)O8 + 2H2O + O + CO2 (Al2Si2)O5 (OH)4 + 4SiO2 + Na2CO3 feldspar kaolinite clay 13 colloidal silica salt Ferromagnesian minerals (i.e., olivine, pyroxenes, amphiboles) also decompose to other varieties of clay with similar byproducts plus colloidal ferric-hydroxide. The soluble salt byproducts are the salts of the oceans and lakes and are largely salts of Na, K, Ca and Mg, which combine with the gasses CO2, SO2 and Cl2 in the sea to form carbonates, sulfates and chlorides. Colloidal silica is transported by groundwater and deposited as a gel in the fractures and pore spaces of clastic sediments as a cementing agent for sand and gravel (Figure 10-20) or deposited as opal or chalcedony in veins, voids or cavities in any rocktype. Colloidal ferrichydroxide is also carried and deposited by groundwater as limonite or goethite (Fe2O3·n H2O), which cements and colors detrital sediments and forms “iron” nodules. Quartz is remarkably stable over a wide range of temperatures and pressures and is stable at the earth’s surface forming sand or pebbles of quartz which are glued, or cemented, together to form sandstone or quartz conglomerate. Sand grains are most commonly quartz, and the cements are most commonly calcite, chalcedony or limonite, but an infinite variety of other minerals and rocks may act as pebbles, sand grains or glue. Although most gems may be recovered from modern river gravels or conglomerates (Figure 10-21) or ancient metaconglomerates, the gems pebbles did not originate in these detrital sediments but were freed, by weathering, from the source rocks where they did form. Most gem minerals tend to be chemically stable and to resist the attack of weather. 14 Shale is very fine-grained sedimentary rock and is composed of clay minerals, which are the products of weathering. Shale is normally thinly layered due to the natural orientation of clay particles which are tiny, mica-like sheets. A wide variety of clay minerals, have a wide range of composition, however, all clays are essentially aluminum hydroxides. Unbalanced electrical charges at the edges of tiny clay layers attract available cations of many other elements becoming the raw materials of metamorphic gem minerals. Metamorphosed shale, always aluminum rich, may contain a greater variety of minerals than any other geological environment, and many gem minerals form in metamorphosed shale, e.g., jade, garnet, iolite, spinel, etc. Chemical sediments are deposited chemically or organically from oceans, lakes, streams or groundwater. Calcium-carbonate (CaCO3), as calcite or aragonite, is by far the most common chemical sediment and has been deposited in huge amounts in shallow seas throughout geologic time. Calcite is the principal constituent of all limestones and marbles which occur in an infinite variety of colors and textures, due to a wide range of impurities. In addition to the iron oxides (i.e., rust) which provide the red, yellow and brown in most rock varieties, limestones may contain large amounts of graphite from decayed marine life, and gray to black limestone is very common. High temperature metamorphism removes graphite as carbon-dioxide (CO2), and the resulting marble tends to be white masses of coarse crystalline calcite. Residual graphite may remain as black or gray streaks or patterns in the marble. Most marine organisms take calcium-carbonate from seawater to form protective shells or armor, and many limestones are composed almost entirely of calcium-carbonate shells or the discarded habitats or skeletons of a wide variety of animals that lived in the sea. Coral is the calcium-carbonate “apartment house” for myriads of tiny marine organisms called polyps. Oysters, 15 mussels, abalone and other shell-fish, which line their shells with calcium-carbonate nacre, yield pearls which are alternating layers of calcium-carbonate and a natural plastic, called conchiolin. Calcium-carbonate is not very soluble and is precipitated from ocean waters, as fast as it is produced by the weathering of plagioclase feldspars, pyroxenes or amphiboles and carried to the sea by rivers and streams. Although the ocean waters are saturated with calcium-carbonate, it is a minor constituent of the soluble salts in sea water. Chalk is fine-grained, rather pure calciumcarbonate deposited as a fine calcite mud or ooze. Calcium-carbonate is also the principal load of groundwater. It is carried in a soluble form as calcium-bicarbonate [CaH2(CO3)2] which, on exposure to the air, releases CO2 gas and water to become insoluble calcium-carbonate (CaCO3). It is calcium-carbonate that makes culinary water “hard” and deposits in pipes and teakettles and white rings on windows, “crystal” goblets and recently washed cars. Calcium-carbonate deposited from ground waters is called travertine (Figure 10-22). It may be deposited from hot spring waters as a highly porous, spongy and fragile rock called tufa, or sinter, or it may be deposited by cold, descending waters in tiny fractures or large veins or in caves as solid, layered, onyx-like rock. This solid, cave travertine is almost always colored yellow to brown by minor iron impurities and is often misnamed “alabaster” when used to make vases, table tops or fireplaces. Hot spring water is often groundwater, heated by hydrothermal water or gasses (e.g., CO3), which may deposit calcite, usually as fine crystals, in association with various ore minerals. In such an environment, a whole family of metal carbonates may form, e.g., siderite (FeCO3), rhodochrosite (MnCO3), smithsonite (ZnCO3), magnesite (MgCO3) and dolomite [CaMg(CO3)2]. 16 This is an isomorphous mineral family ( i.e., same trigonal crystal structure, but different compositions) with similar properties. Rhodochrosite, containing manganese as an essential constituent, is always rose pink and smithsonite, which usually contains enough copper ions to color it pale blue-green, are both sometimes cut and polished as ornamental stone. Aragonite is an orthorhombic polymorph of calcite (i.e., same composition but different crystal structure) and will revert slowly to calcite under normal surface conditions of temperature and pressure. With slightly higher pressure or lower temperature, aragonite becomes the stable, polymorph, in some cave deposits and the calcium-carbonate layers in pearls or shells. Strontianite (SrCO3), witherite (BaCO3) and cerussite (PbCO3) have large cations and the same orthorhombic structure as aragonite. i.e., they are isomorphs of aragonite. Metamorphic Rocks Metamorphic rocks (Figure 10-23) are secondary rocks derived from any previously existing rock which is subjected to elevated temperatures and/or pressures sufficient to alter its texture, fabric, or mineral combinations but insufficient to bring about melting and its return to a magma. Metamorphism is normally associated with subduction zones (Figure 10-5) where surface rocks of all kinds are carried into the mantle as part of the subducting plate. In the zone of mountain building, where sedimentary formations are deformed by compressional forces, temperature increases with depth, and pressure is directional (sheer pressure) near the surface grading to uniform pressure, or uniform compression, at depth. Any chemical system (i.e., combination of elements) will form one combination of minerals, under one specific range of temperature and pressure, and perhaps another combination 17 of minerals, under another set of temperature-pressure conditions. This is metamorphism, and many gem minerals are formed in this way. Several types of metamorphism are defined by a range of temperature and a range of pressure, and metamorphic “facies” are defined by the combination of minerals which the temperature, pressure and chemistry produce. At shallow depth, where the under riding plate, carrying ocean sediment, goes beneath the overriding plate, temperature is quite low but shearing pressure is high (blueschist metamorphism). Under these conditions, ocean salts rich in sodium, may be added to the mix of ocean clay sediments and “shear” minerals are formed, like jadeite [NaAl(SiO3)2] and glaucophane [Na2Mg3Al2(Si4O11)2(OH)2]. In the main mountain building system, where sedimentary rocks are highly deformed and masses of granitic magmas are generated, temperatures and pressures are low near the surface becoming greater at depth (dynamothermal metamorphism). At moderate depths, shearing pressures and moderate temperatures may alter a stable mineral combination in ocean sediments (e.g., sand, clay, iron oxides) to form a different set of minerals (e.g., mica, chlorite, garnet) and the oriented texture of a schist (Figure 10-24A) without changing the total chemical composition of the overall rock. At greater depth (i.e., higher temperature and more uniform pressure), the mica and chlorite may become unstable and change to feldspar and amphibole or pyroxene, and the oriented, flaky schistose fabric of the rock changes to the granular, deformed layering of a gneiss (Figure 1024B) or, at even greater depth, the coarse granular texture of a granite-like rock. Almandite garnet [Fe3Al2(SiO4)3], staurolite [Fe2Al9O6(SiO4)4(OH)2], iolite [Mg2Al3(Si5Al)O18] and kyanite [Al2SiO5] are common gem minerals of schists and gneisses. Where molten rock intrudes solid rock at shallow depth (contact metamorphism), the 18 temperature is high and pressure low, heat from the molten rock, conducted to the solid wallrock, may alter its texture and recombine its elements to new mineral combinations, without altering its total chemistry (Figure 10-25). Gem minerals like grossular garnet [Ca3Al2(SiO4)3], andalusite [Al2SiO5} and idocrase [Ca10(Mg,Fe)2Al4(Si2O7)2(SiO5)5(OH)4] are metamorphic contact minerals. Notice that all the mineral formulas for metamorphic minerals in the paragraphs above contain only the eight most abundant elements, plus hydrogen from water. These metamorphic minerals reflect the composition of the original sedimentary or igneous rocks from which they formed, without addition or subtraction of foreign elements. When volatile-rich molten rock, perhaps a pegmatite magma, intrudes a solid rock, however, the “country rock” may receive more than heat from the magma. It may be infiltrated with gasses and fluids, from the magma, containing trace elements to be added to minerals of the new mineral combinations. This form of contact metamorphism is called metasomatism and may yield gem minerals like tourmaline [Na(Mg, Fe, Li, Al)3Al6(SiO3)6(BO3)3(OH,F)4] and lazurite [(Na,Ca)8Al6Si6O24(SO4,S,Cl)2]. In metasomatic minerals we commonly see the addition of gasses (SO2, S, Cl, F) and trace elements (Li, B), from cooling magmas, and residual salt brines (NaCl) from marine sediments. Metamorphism of shale, composed mostly of clays [e.g., Al2Si2O5)(OH)4 ], yields metamorphic minerals rich in aluminum. Carbonate sediments, composed mostly of calcite and dolomite [i.e., CaCO3 and CaMg(CO3)2], yield metamorphic minerals rich in calcium and magnesium. Basaltic rocks, rich in mafic minerals [e.g., pyroxene CaMg(SiO3)2 and amphibole Ca5(Mg,Fe,Al)5(Si4O11)2(OH)2], yield metamorphic minerals rich in magnesium, iron and calcium. Limestone (CaCO3) containing an aluminum-hydroxide (e.g., bauxite) may yield corundum 19 (Al2O3), as a metamorphic mineral, and if the limestone is magnesian calcite, spinel (MgAl2O4) may result. Corundum and spinel must form in a silica-poor environment 20