Topic 11 – Group 2 & Group 7
advertisement

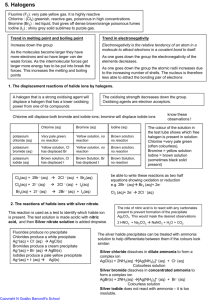
Topic 14b – Group 7, the Halogens Revision Notes 1) Trends in physical properties of the elements 2) At room temperature, Cl2 is a pale green gas, Br2 is a brown liquid, I2 is a blue-black solid. In Group 7, boiling point increases down the group because the molecules have more electrons and, therefore, stronger van der Waal’s forces which require more energy to overcome them Electronegativity is the ability of an atom to attract the electrons in a covalent bond Down Group 7, the shared electrons are further from the nucleus and more shielded. There is a weaker attraction for the bonding electrons down the Group Trends in chemical properties of the elements The Group 7 elements are oxidising agents which gain an electron when they react The ability to oxidise decreases down Group 7 This can be shown by halogen displacement reactions where elements higher up the group will displace elements further down the group This can be done with chlorine dissolved in water or by bubbling chlorine gas through NaBr(aq) or NaI(aq) Cl2 + 2NaBr 2NaCl + Br2 Pale green solution Yellow solution/orange in hexane Cl2 + 2NaI 2NaCl + I2 Pale green solution Brown solution/purple in hexane Br2 + 2NaI 2NaBr + I2 Orange solution Brown solution/purple in hexane These equations can also be written in ionic form Cl2 + 2Br- 2Cl- + Br2 Cl2 + 2I- 2Cl- + I2 Br2 + 2I- 2Br- + I2 3) Down the group the outer electron is further from nucleus and more shielded so gained less easily Trends in properties of the halides The halides (chlorides, bromides and iodides) are reducing agents which lose an electron when they react The ability to act as a reducing agent increases down the Group Down the group the outer electron is further from nucleus and more shielded so lost more easily This can be shown by the reactions of the solid halides with concentrated sulphuric acid Sodium halide Observations Products Type of reaction NaF Steamy fumes HF(g) Acid-base (F- acting as base) NaCl Steamy fumes HCl(g) Acid-base (Cl- acting as base) NaBr Steamy fumes Colourless gas Brown fumes HBr(g) SO2 Br2 Acid-base (Br- acting as base) Redox (reduction product of H2SO4) Redox (oxidation product of Br-) NaI Steamy fumes Colourless gas Yellow solid Smell of bad eggs Black solid/purple fumes HI(g) SO2 S H2S I2(s/g) Acid-base (I- acting as base) Redox (reduction product of H2SO4) Redox (reduction product of H2SO4) Redox (reduction product of H2SO4) Redox (oxidation product of I-) 4) Iodide ions, I-, can reduce the S from oxidation state +6 in H2SO4 to +4 in SO2 then to 0 in S and finally to -2 in H2S Bromide ions, Br-, can reduce the S from oxidation state +6 in H2SO4 to +4 in SO2 Fluoride and chloride cannot reduce the S in H 2SO4 under these conditions Reactions of Cl-, Br- and I- with Silver Ions and Ammonia Solution Chloride ions, bromide ions and iodide ions produce coloured precipitates with acidified silver nitrate solution. Fluoride ions do not produce a precipitate with AgNO 3 Ag+(aq) + Cl-(aq) AgCl(s) White precipitate Ag+(aq) + Br-(aq) AgBr(s) Cream precipitate Ag+(aq) + I-(aq) 5) AgI(s) Yellow precipitate AgCl(s) dissolves in dilute ammonia solution, NH 3(aq) AgBr(s) dissolves in concentrated ammonia solution but not in dilute ammonia. AgI(s) does not dissolve, even in concentrated ammonia solution Redox Reactions of Group 7 Elements Chlorine undergoes a redox reaction with water. This reaction is used in water purification to kill bacteria Cl2(g) + H2O(l) HCl(aq) + HOCl(aq) The benefits to health of water treatment outweigh the risks associated with handling toxic chlorine gas Chlorine reacts with cold, dilute sodium hydroxide solution. This reaction is used to make household bleach Cl2(g) + 2NaOH(aq) NaCl(aq) + NaClO(aq) +H2O(l) NaClO is sodium chlorate(I) which is the active ingredient in bleach In both of these reactions the oxidation state of Cl changes from 0 in Cl 2 to –1 in Cland +1 in OCl-. Cl is simultaneously oxidised and reduced. This is an example of disproportionation Topic 14a – Group 2, the Alkaline Earth Metals Revision Notes 1) Trends in Physical Properties Down Group 2 (Be-Ba) a) Atomic radius b) Increases down the group Extra electron shell, outer electron further from nucleus and more shielded Increased nuclear charge outweighed by greater shielding and distance 1st ionisation energy c) Decreases down the group Extra electron shell, outer electron further from nucleus and more shielded Increased nuclear charge outweighed by greater shielding and distance Melting & boiling points Decrease down the group Metallic bonding gets weaker down the group. Ionic radius of metal ions increases down group so charge density decreases. Weaker attraction between metal ions and delocalised electrons 2) Trends in Chemical Properties Down Group 2 (Be-Ba) a) Reactivity with Water Reactivity increases down the group Extra electron shell, outer electrons further from nucleus and more shielded so lost more easily. Increased nuclear charge outweighed by greater shielding and distance Mg reacts slowly with cold water. Calcium reacts more vigorously Ca(s) + 2H2O(l) Ca(OH)2(aq) + H2(g) Mg reacts rapidly with steam Mg(s) + H2O(g) MgO(s) + H2(g) b) Solubility of Sulphates Solubility of group 2 sulphates decreases down the group i.e. BaSO4 is the least soluble group 2 sulphate BaCl2(aq) is used to test for sulphates. A white precipitate is produced Ba2+(aq) + SO42-(aq) BaSO4(s) Barium sulphate is used for X-ray imaging and CT scans. It is most often used in imaging of the oesophagus. It is not readily absorbed by the body and absorbs Xrays more strongly than lighter atoms c) Solubility of hydroxides Solubility of group 2 hydroxides increases down the group. Mg(OH) 2 is sparingly soluble i.e. only a small amount dissolves Group 2 hydroxides are alkaline and can be used to neutralise acids Calcium hydroxide, Ca(OH)2, can be used to reduce soil acidity in agriculture Magnesium hydroxide, Mg(OH)2, is found in milk of magnesia. This is used to treat indigestion by neutralising excess HCl in the stomach (the Mg(OH)2 acts as an antacid)