SUPPLIMENTAL SHEET FOR CHEMICAL PRICIPLES IN THE
advertisement

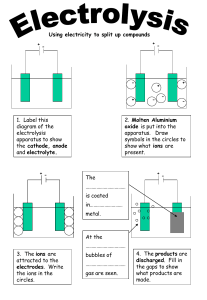
SUPPLIMENTAL SHEET FOR CHEMISTRY 121 LAB MANUAL, OAKTON COMMUNITY COLLEGE (SLOWINSKI) Experiment 2 due next lab period The completed “Advanced Study Assignment” (page 9) is due with the experiment. 1. Measure distances in the diagram from the “point of application” and calculate Rf values. NOTE: 1.0 X 10 -6 g = 1.0 µg DO NOT RETURN ANYTHING TO THE STOCK BOTTLES. 1. Deposit waste in the correct waste bottle in the hood. Check the experiment # on the bottle. 2. Record your sample number. 3. Use the filter paper in your desk drawer to test for ions. 4. DO NOT TAKE MORE THAN 1 mL OF SOLUTION. 5. The paper is already 19cm x 11cm. HANDLE THE PAPER WITH FORCEPS. DO NOT TOUCH TREATED PAPER. 6. You will analyze 1 unknown. The same unknown is done at two places. 7. Let the experiment run until the eluting solution is 2/3 of the way to the top of the chromatography paper (about 60 minutes) – not 75 minutes indicated on bottom of page 5. 8. There are possible 5 ions (Ag+, Co2+, Cu2+, Fe2+, Hg2+ ). Your unknown has 3 of those ions. Determine the 3 ions you have to the best of your ability during the first laboratory period. However, you may store the chromatogram in your lab drawer to be reexamined during the next lab period. The experiment is due during the next lab period. BEFORE YOU LEAVE, WASH YOUR HANDS.