Characteristics of Gases
advertisement

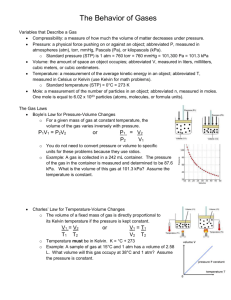
Ch. 12 Gas Laws Outline Characteristics of Gases 1. Gases expand to fill the volume of whatever container they are in. 2. Gases are mostly empty space - the molecules are relatively far apart. 3. Gases always form homogeneous solutions. 4. Gases are easily compressed. Substances that exist as gases at room temperature: - nonmetals with low molecular masses (ex: H2,N2,O2,F2,Cl2) - noble gases - molecular compounds with low molecular masses are often gases (ex: CO2,NH3,CH4) Metallic or ionic substances are NOT gases at room temperature. vapor - molecules of a normally liquid or solid substance that have entered the gas phase. Pressure –Force/Area; amount of force per unit of area. Units of pressure: kilopascals (kPa), atmospheres (atm), millimeters of mercury (mmHg), torr (a torr and a mmHg are the same thing) standard pressure - the average atmospheric pressure at sea level. Standard pressure is defined as: 760 mm Hg = 760 torr = 1 atm = 101.3 kPa Temperature Kelvin temperatures must be used when using the gas laws. Temperature conversion: K = ºC + 273 STP - Standard Temperature and Pressure STP = 0ºC and 1 atm The Gas Laws A. Boyle's Law - the volume and pressure of a sample of gas are inversely proportional (if temperature remains constant). Read section 12-2 pgs 299-303 Boyle's Law equation: P1V1 = P2V2 (P1 and V1 are initial (beginning) pressure and volume; P2 and V2 are final pressure and volume.) B. Charles' Law - the volume of a sample of gas is directly proportional to its Kelvin temperature (if pressure stays constant). Read section 12-3 pages 303-309 Charles' Law equation: V1/T1 = V2/T2 (V1 and T1 are initial (beginning) volume and temp; V2 and T2 are final volume and temp.) C. Temperature and Pressure Law - the pressure of a sample of gas is directly proprtional to its Kelvin temperature (if volume stays constant). Read 12-4 pages 310 Pressure-Temperature law equation: P1/T1 = P2/T2 Gas Laws Outline Page 2 E. Combined Gas Law Combines Boyle’s, Charles’, and pressure-temp laws. Sec 12-5 pgs 311-313 F. Ideal Gas Law - relates the pressure (P), volume (V), # of moles (n), and Kelvin temperature (T) of a sample of gas. Read 12-13 pages 326-328 Ideal Gas Law equation: PV = nRT R is the gas constant. The value of R that you use in a problem depends upon what the other units in the problem are. The most commonly used values of R are: 0.0821 (L*atm)/(mol*K) or 8.31 (dm3*kPa)/(mol*K) See Molar mass derivation in notes. G. Dalton’s Law of Partial pressures (sec 12-9,pg318))– The total pressure of a mixture of gases is equal to the sum of the pressures each individual gas would exert in the same volume. H. Graham’s Law of Diffusion (Sec12-10,pg 320)– The rate of one gas diffusing compared to another gas is equal to the inverse of the square roots of their molar masses (or densities). Read over Section 12-11, pg322-323, Kinetic Theory and the Gas Laws