Gas Laws
advertisement

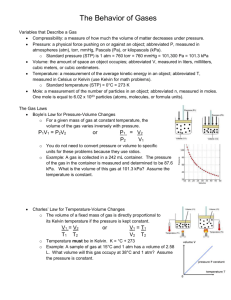
Gas Laws A. Characteristics of Gases • Gases expand to fill any container. – random motion, no attraction • Gases are fluids (like liquids). – no attraction • Gases have very low densities. – no volume = lots of empty space B. Characteristics of Gases • Gases can be compressed. – no volume = lots of empty space • Gases undergo diffusion & effusion. – random motion C. Temperature • Always use absolute temperature (Kelvin) when working with gases. ºF -459 ºC -273 K 0 C F 32 5 9 32 212 0 100 273 373 K = ºC + 273 D. Pressure • KEY UNITS AT SEA LEVEL 101.325 kPa (kilopascal) 1 atm 760 mm Hg 760 torr 14.7 psi N kPa 2 m E. STP STP Standard Temperature & Pressure 0°C 273 K -OR- 1 atm 101.325 kPa Units STP is Standard Temperature and Pressure. STP is 0 °C and 1 atm of pressure. Gases properties can be compared using STP as a reference. Temperature: Celsius and Kelvin 0 ◦C = 273 K Pressure Units • 1 atm • 760 mmHg • 101.3 kPa • 760 torr Using dimensional analysis to convert pressure units • What is 475 mm Hg expressed in atm? 475 mmHg 1 atm = 0.625 atm 760 mm Hg Charles's Law Charles's Law can be stated as the volume occupied by any sample of gas at a constant pressure is directly proportional to the absolute temperature. • • • • • • • • V is the volume T is the absolute temperature (measured in Kelvin) Charles's Law can be rearranged into: V1 / T1 = V2 / T2 V1 is the initial volume T1 is the initial temperature V2 is the final volume T2 is the final temperature Sample Problem You have a gas that has a volume of 2.5 liters and a temperature of 250 K. What would be the final temperature if the gas has a volume of 4.5 liters? V1 / T1 = V2 / T2 V1 = 2.5 liters T1 = 250 K V2 = 4.5 liters T2 = ? Solving for T2, the final temperature equals 450 K. So, increasing the volume of a gas at constant pressure from 2.5 to 4.5 liters results in a temperature increase of 200 K. Important: Charles's Law only works when the pressure is constant. Note: Charles's Law is fairly accurate but gases tend to deviate from it at very high and low pressures. Boyle's Law • Boyle's Law states the volume of a definite quantity of dry gas is inversely proportional to the pressure, provided the temperature remains constant. • • • • • Mathematically Boyle's law can be expressed as P1V1 = P2V2 V1 is the original volume V2 is the new volume P1 is original pressure P2 is the new pressure Sample Problem • Suppose you have a gas with 45.0 ml of volume and has a pressure of 760.mmHg. If the pressure is increased to 800mmHg and the temperature remains constant then according to Boyle's Law the new volume is 42.8 ml. • (760mmHg) * (45.0ml) = (800mmHg) * (V2) 800 mmHg 800 mmHg V2 = 42.8ml To solve for V2, divide both sides by 800 mmHg. Cancel 800 mm Hg on the right side. Then multiply 760 by 45.0. Divide your answer by 800 and you should get 42.8 Gay-Lussac’s Law • P and T are directly Proportional P1 = P2 T1 T2 or P1T2 = P2T1 Sample problem The pressure inside a container is 770 mmHg at a temperature of 57 C. What would the pressure be at 75 C? P1= 770 mmHg T1 = 57°C T2= 75°C P2 = ? Combined Law • The combined gas law is a combination of Boyle's Law and Charles's Law; hence its name the combined gas law. Since for a given amount of gas there is a constant then we can write-P1V1 / T1 = P2V2 / T2. • • • • • • P1 is the initial pressure V1 is the initial volume T1 is the initial temperature (in Kelvin) P2 is the final pressure V2 is the final volume T2 is the final temperature (in Kelvin) • For example if you have 4.0 liters of gas at STP, and you want to know the volume of the gas at 2.0 atm of pressure and 30 °C, the equation can be setup as follows: (1.0)(4.0) / 273 = (2.0)(V2) / 303 (V2)(2)(273) = (1)(4)(303) V2 = 2.2 Therefore the new volume is 2.2 liters •