Solubility and Concentration Review
advertisement

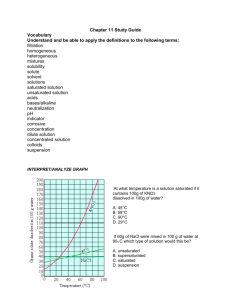
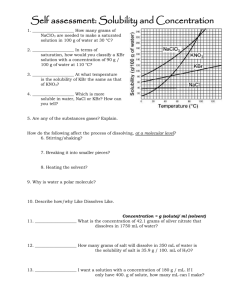
Name: _____________________________ Score: _____/21 Self-assessment: Solubility and Concentration 1. – 2. There are two parts to a solution. Name and describe each. 3. – 4. Draw a water molecule and explain why it is a polar molecule. 5. – 6. Draw and describe a hydrogen bond. 7. Describe how/why “Like Dissolves Like.” How do the following affect the process of dissolving, at a molecular level? 8. Stirring/shaking? 9. Breaking it into smaller pieces? 10. Heating the solvent? 11. If a solution is holding the maximum amount it can it is called _________________________. 12. If it’s holding more than it usually can it is called _______________________________. 13. If it’s holding less than the maximum amount it is called _______________________________. When I look at a solution, it will look the same no matter if it’s unsaturated, saturated or super saturated. 14. What can I do to tell which one it is. 15. -17. What will happen if it is: Unsaturated Saturated Supersaturated: Concentration = g (solute)/ ml (solvent) 18. ____________________ What is the concentration of 42.1 grams of silver nitrate that dissolves in 1750 mL of water? 19. ____________________ I have a solution of copper chloride with a concentration of 3.47 g/ml. How much copper chloride do I need to evaporate to get 300.00 g out of it? 20. ____________________ How many grams of salt will dissolve in 350 mL of water is the solubility of salt is 35.9 g / 100. mL of H2O? 21. ____________________ I want a solution with a concentration of 180 g / mL. If I only have 400. g of solute, how many mL can I make? Answer Key__ Score: _____/21 Self assessment: Solubility and Concentration Name: __ 1. – 2. There are two parts to a solution. Name and describe each. Solute (what’s being dissolved) Solvent (what’s doing the dissolving) 3. – 4. Draw a water molecule and explain why it is a polar molecule. “Uneven distribution of electrons”…electrons not shared equally…oxygen pulls on them a little more than hydrogen, so one side has a little positive charge (+) and one side is a little negative (-) 5. – 6. Draw and describe a hydrogen bond. Bond BETWEEN water molecules (not within 1 molecule) from little bit positive end of one to little bit negative end of the other 7. Describe how/why Like Dissolves Like. Polar things will dissolve in polar solvents, nonpolar solutes will dissolve in nonpolar solvents. Because the charges in a polar molecule will pull apart the other if it also has charges. (polar will pull apart polar, but not nonpolar) How do the following affect the process of dissolving, at a molecular level? 8. Stirring/shaking? Moves molecules away, so more room for interactions with solute. 9. Breaking it into smaller pieces? More surface area for interactions between water and solute molecules. 10. Heating the solvent? Molecules of solute and solvent move faster and interact more 11. If a solution is holding the maximum amount it can it is called _ 12. If it’s holding more than it usually can it is called _ saturated_. supersaturated _. 13. If it’s holding less than the maximum amount it is called _ unsaturated _. When I look at a solution, it will look the same no matter if it’s unsaturated, saturated or super saturated. 14. What can I do to tell which one it is. Add more powder: 15. - 17. What will happen if it is: Unsaturated Saturated powder will dissolve powder will not dissolve, original amount will fall to bottom powder will not dissolve, more than original amount will fall to the bottom Supersaturated: Concentration = g (solute)/ ml (solvent) .0241 g/mL__ What is the concentration of 42.1 grams of silver nitrate that 18. _ dissolves in 1750 mL of water? 42.1 1750 = .0240571 86.5 ml_ I have a solution of copper chloride with a concentration of 19. __ 3.47 g/ml. How much copper chloride do I need to evaporate to get 300.00 g out of it? 3.47 = 300 x (mult by x) 3.47(x) = 300 (div by 3.47) x (mult by x) 300 3.47 x = 86.45533141 20. __ = 130 g___ How many grams of salt will dissolve in 350 mL of water is the solubility of salt is 35.9 g / 100. mL of H2O? 35.9 100 = x 350 (cross muliply) 35.9 x 350 = 100 x x = 125.65 21. __ 12565 = 100 x 2.2 mL __ I want a solution with a concentration of 180 g / mL. If I only have 400. g of solute, how many mL can I make? 180 = 400/x 180x = 400 x = 400/180 (mult by x) (div by 180) x = 2.22222222