4.2 - Representing Molecular Compounds
advertisement

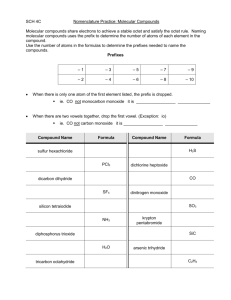
4.2 Representing Molecular Compounds (Page 152) Name:____________________________ _________________________ are usually composed of two or more different _____________________. The elements within a molecular compound __________________________. A water molecule with ________ and hydrogen molecules sharing electrons Draw the Lewis structure for this molecular compound below: Molecular compounds feature ________________________ between ____________________ and are often referred to as _________________________________. Molecules are __________________ particles composed of ____________________ atoms joined together. Diatomic Molecules - Naming Binary Molecular Compounds A binary molecular compound is composed of _________________ joined by one or more covalent bonds. __________________ indicate the _________________ of atoms of each element in a molecule of the compound. Things to know prior to naming: Step 1: Use prefixes to indicate the number of both atoms in the compound (given in the chemical formula) Step 2: The name of the second element ends in –ide. *Write the elements in order of how the appear in the chemical formula* Example 1: Name SO3 Example 2: Name N2O4 Writing Chemical Formulas for Molecular Compounds Example: Write the chemical formula for disulfur dinitride Step 1: Write down the symbol of the elements with the subscripts indicated by the prefixes. Note: (A simplified ratio can result in a different molecular compound. For example, NO2 and N2O4 are different compounds!) Common names of molecular compounds Practice: Name each of the following compounds. a) BrF __________________________ b) NO __________________________ c) CO2 __________________________ d) SO3 __________________________ e) As2O3 __________________________ f) PBr5 g) Br2O h) P2O3 i) CO j) P4O10 __________________________ __________________________ __________________________ __________________________ __________________________ Write the chemical formula for each of the following compounds. a) Iodine monofluoride _____________ f) carbon tetrafluoroide b) Arsenic trichloride _____________ g) hydrogen chloride c) sulfur dioxide _____________ h) dichlorine monoxide d) phosphorus pentabromide _____________ i) dichlorine pentoxide e) nitrogen trichloride _____________ j) sulfur hexafluoride Homework Read Pages 152-158 and Answer Q’s 1-3, 6 on Page 158 Read Pages 159-168 and Answer Q’s 1, 3, 5, 6 on Page 168 _____________ _____________ _____________ _____________ _____________