Exam 1 Answer Key
advertisement

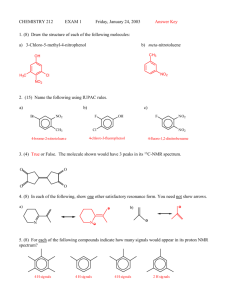
CHEMISTRY 212 EXAM 1 Thursday, January 18, 2007 Answer Key **1. (16) For each of the following compounds indicate how many signals would appear in both its carbon and proton NMR spectra? OH H: 5 signals; C: 3 signals H3 C O CH3 H3 C O CH3 H: 2 signals; C: 2 signals H: 4 signals; C: 4 signals H: 3 signals; C: 4 signals **2. (13) Show reasonable products from the following reactions. Draw the stereochemistry of the product(s) as appropriate. a) NO2 NO2 b) Cl Br H3C Br CH3 Me NO2 + + Cl Cl Br K+ -OC(CH3 )3 Br + H H **3. (5) Which of the following compounds has the spectral characteristics described? Proton NMR: 1.75 ∂ (singlet); 2.00 ∂ (triplet); 3.75 ∂ (triplet). O Cl Cl O Cl O O Cl H **4. (8) For the two molecules shown, describe which method (proton NMR or carbon NMR) would allow the clearest distinction between them. Explain briefly but clearly what would be observed in both spectra for both molecules, with appearance and approximate positions of the peaks. Both molecules would have almost identical H and C-NMR spectra - showing: 1H : one singlet at ~1 and one singlet at ~7-8 13 C : a singlet at ~30 and two singlets at ~130 The only distinguishing feature would be the relative areas of the peaks in their proton NMR sp ectra. This, the molecule on the left would have a ratio of 6 : 1 for the methy ls and CH's whereas the molecule on the right would have a 3 : 2 ratio. This would be easily observed. **5. (2.5) True or False. A position further to the left in an NMR spectrum is called deshielded. **6. (4) The two general types of molecule required for a Diels-Alder reaction are called? Conjugated Diene Dienophile **7. (3) Sharp, medium strength peaks at ~1600 and at 1500 cm-1 in an IR spectrum would indicate ? A. A carboxylic acid B. An alkene C. An aromatic ring D. An amine E. A ketone **8. (6) Which of the following gives only one product upon E2 reaction with KOH? b) a) c) Br H3C CH3 d) CH2Br Br Cl **9. (7.5) For each of the following compounds, indicate by yes or no whether or not they contain a conjugated system. a) b) c) Yes No No **10. (4.5) What splitting pattern (singlet, doublet, triplet, quartet, quintet, sextet) is observed in the 1H-NMR spectrum for the hydrogen on the indicated carbon? sextet due to 5 adjacent H's, none of which is the same as the H of concern **11. (7.5) At what locations (give numbers) would you expect to see peaks in the IR spectrum of the following molecule? O O HO OH Carboxylic acid OH str. at 3400-2400 cm -1 CH str. aromatic at 3100 cm -1 CH str. alkyl at 2900 cm -1 C=C str. aromatic at 1600 and 1500 cm -1 C=O str. at 1700 cm -1 ; CH bend aromatic at 800 cm -1 **12. (3) True or False? Using only the functional groups & their locations discussed in class, it would be easy to tell the difference between the following two compounds using only their infrared spectra? N O O H **13. (6) The 2 molecules shown below could be distinguished easily by their 13C-NMR spectra. Explain briefly, but clearly, why. Cl Br Cl Cl Cl Br Br Br due to this plane of symmetry there are 3 C signals due to this plane of symmetry there are 4 C signals **14. (6) Draw the different alkene products which would result from the following reaction. Additionally, circle the major product. Br NaNH2 E&Z + **15. (3) Why do you suppose that accidental overlap of signals is much more common in 1H-NMR than in 13C-NMR? Explain briefly. The main reason is that there is splitting of peaks in H-NMR (doublets, triplets etc.), which is not observed in C-NMR (as we've studied it). This gives more opportunities for overlap of peaks. **16. (6) Which of the following molecules displays 3 signals in its 13C NMR spectrum? **17. (4.5) Give 3 key requirements for the internal standard (TMS) used for NMR spectra. Must be: Easily removed; unreactive chemically; contain many identical H's ; contain C's and H's which are different from the mainstream allowing them to be distinguished from the C's and H's in most organic compounds. **18. (19.5) For each of the following, circle any answer to the right that is correct (note: there could be more than one correct answer in some cases). a) + polar protic solvent / poor nucleophile Br ii) S N 1 b) + CH 3 O- K+ Cl c) i) Inversion of Configuration + EtOH ii) Racemic Mixture formation Cl H i) S N2 ii) S N 1 Cl O H d) i) S N2 + LiCN i) 4 times the reaction rate if double the concentration of both nucleophile and alkyl halide ii) 2 times the reaction rate if double the concentration of both nucleophile and alkyl halide e) + Br NaOEt i) No S N2 reaction ii) S N 1 reaction iii) E2 reaction DMSO CH 3 **19. (7.5) Circle the reactant in each of the following pairs which is more nucleophilic. a) I and b) F CH4 and c) Cl NH3 and OH2 **20. (7.5) Circle the species in each of the following pairs which is a better leaving group. a) I and b) F CH3 and c) Cl N2 and **21. (5) Circle the species in each of the following pairs which is the stronger base. a) H2 N and HO b) H2 O and Cl I gave credit to either of these since I was unsure that you could know the answer from our class discussion **22. (2.5) The anti-periplanar conformation is required for an E2 reaction because: A. Planar molecules are more stable B. E2 reactions give products with an anti arrangement C. This conformation leads to more stable (E) alkenes D. This conformation allows the base and leaving group to be far apart **23. (2.5) There are two key general areas in an infrared spectrum - name them. Functional group region and fingerprint region OH