bodeneq
advertisement

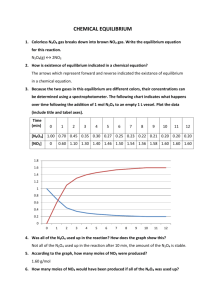
Bodenstein equilibrium Solutions 1) For the gas-phase reaction 2 HI H2 + I2 , the equilibrium constant is K = pHpI/pHI2 . If the system contains only HI initially, at a pressure p0, and if the fraction of HI that decomposes by the time the reaction reaches equilibrium, then, at equilibrium pHI = (1 – )p0 , and pH = p0/2 = pI . The latter result reflects the fact that H2 and I2 are formed in equal proportions, with a half mole of each formed for every mole of HI that decomposes. Thus the equilibrium constant is p p (p0 /2)(p0 /2) 2 p0 2 2 , K H 2I pHI [(1 )p0 ]2 4(1 ) 2 p0 2 4(1 )2 as was to be proved. 2) Computing the equilibrium constants is a simple matter of plugging in numbers, such as: K 2 (0.2408)2 2 2 = 0.02515 . 4(1 ) 4(1 0.2408) The computed values are shown in the table below. temp (°C) T(K) 1/T(K) K 508 781 0.00128 0.2408 0.02515 487.2 760.2 0.00132 0.2340 0.02333 443 716 0.00140 0.2198 0.01984 427 700 0.00143 0.2157 0.01891 410 683 0.00146 0.2100 0.01767 393 666 0.00150 0.2058 0.01679 374 647 0.00155 0.2010 0.01582 356 629 0.00159 0.1946 0.01459 328 601 0.00166 0.1885 0.01349 302 575 0.00174 0.1815 0.01229 283 556 0.00180 0.1787 0.01184 To download a worked spreadsheet, go to: http://webserver.lemoyne.edu/faculty/giunta/classicalcs/bodenstein.xls ln K –3.683 –3.758 –3.920 –3.968 –4.036 –4.087 –4.146 –4.227 –4.306 –4.399 –4.437 3) The relationship of the equilibrium constant to standard enthalpy and entropy of reaction is G H S . K exp exp exp RT RT R Taking the natural log of both sides gives: ln K = –(∆H°/R)(1/T) + ∆S°/R . If ∆H° and ∆S° are temperature independent (an assumption to which we will return below), then a plot of ln K vs. 1/T (called a van’t Hoff plot) should yield a straight line whose slope m is equal to –∆H°/R and whose y-intercept b is equal to ∆S°/R . Thus, we obtain ∆H° and ∆S° from the fit of the plotted data: ∆H° = –mR and ∆S° = bR . To make the van’t Hoff plot, we must convert the temperatures to absolute (Kelvin) temperatures and find their reciprocals and we must take the natural log of the equilibrium constant, all of which is done in the table displayed above with exercise 2. The plot, including the equation of the best-fit straight line, is below HI decomposition equilibrium -3.6 ln K -3.8 -4.0 -4.2 -4.4 ln K = - 1.8485 - 1474/T R^2 = 0.979 -4.6 0.0012 0.0014 0.0016 0.0018 1/T(K) So ∆H° = –mR = –(–1474 K)(8.3145 J mol–1 K–1) = 1.23x104 J mol–1 , ∆H°= 12.3 kJ mol–1 , and ∆S° = bR = (–1.8485)(8.3145 J mol–1 K–1) = –15.4 J mol–1 K–1 . NIST thermodynamic tables yield ∆H° = 12.3 kJ mol–1 and ∆S° = –16.0 J mol–1 K–1 at 393°C (near the mid-point of the range of Bodenstein’s data). This agreement is extremely good, attesting to the high quality of Bodenstein’s measurements. Finally, note that the “scatter” of experimental data around the best-fit line does not appear to be random. The data appear to define a curve rather than a straight line. A curved van’t Hoff plot is an indication that the standard enthalpy and entropy of reaction are not strictly independent of temperature. Strictly speaking, ∆H° and ∆S° for a given temperature can be derived from the straight line that is tangent to the ln K vs. 1/T curve at that temperature.