7.2Equilibrium Constant Calculation.4u
advertisement

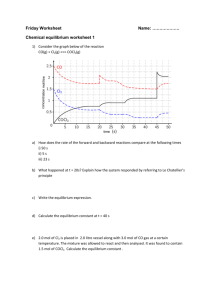
Equilibrium Constant Calculation Consider the equilibrium: A(g) + 2 B(g) <-> C(g) If there were 6.0 mol of A and 4.0 mol of B placed into a 2.0 L container, after a period of time an equilibrium was reached and there was 1.0 mol of C. Calculate the K value. Solution: Step 1 – change moles to concentrations (mol/L) Step 2 – use an ICE table of calculation A(g) + 2 B(g) <-> C(g) Initial 3.0 2.0 0.0 change___________-0.5___-1.0____+0.5________________ equilibrium ___ ___ ___ Step 3 – Calculate the equilibrium line from the concentration that must have been removed from the reactant. (A and C are in a 1:1 ratio and B and C are in a 2:1 ratio) Step 4 - put the equilibrium concentrations into the equilibrium constant expression. K= [C]___ [A] [B]2 K= ____ K=