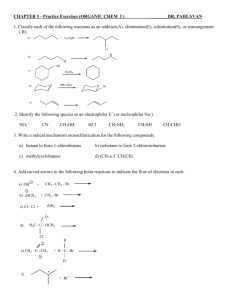
c. Using pKa`s of Acids and Bases to Investigate the Stabilities of
advertisement

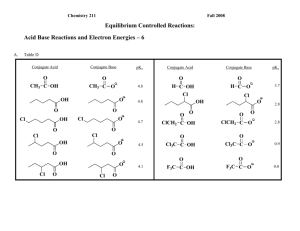
Fall 2007 Chemistry 211 Class Group Activity #10: Acid Base Reactions & Energy Relationships - 4 Summary of Class Discussion A. Table C Conjugate Base Conjugate Acid - CH3 CH3 CH3 CH2 CH2 CH2 CH2 CH HC CH HC C + CH3 CH2 NH3 - + NH CH3 C CH3 C H CH3 O C H CH3 C CH3 + ~40 25 10 5 O -2 CH CH3 O CH3 + CH3 ~42 H H H CH CH3 - CH3 CH2 NH2 NH2 H + O pKa NH CH3 CH3 C - 7.2 CH3 C N -10 B. Based upon the data in Table C, Within each pair of ions below, choose the one with the highest energy electron pair: H highest energy electron pair - H - .. .. N .. .. .N. .. highest energy electron pair Explain the reasoning behind your choices. 1. Difference between members of each pair of ions. In both pairs the comparison is between an ion with a lone pair of e -'s on a negatively charged atom that is involved in a single bond vs. a similar atom involved in a double bond. 2 Acid-Base Rxns & Electron Energy-4 2. Conclusions from Table C data: The higher the bond order (triple bond > double bond > single bond) between the atom holding the proton to be lost and the next atom in the compound, the more acidic is the proton. This is true whether the acid is C, N or O (Table C: pKa's ~42 vs. ~40 vs. 25, 10 vs. 5 vs. -10 and -2 vs. -7.2) or is positive (Table C: pKa's 10 vs. 5 vs. -10 and -2 vs. -7.2) or neutral (pKa's ~42 vs. ~40 vs. 25). Least Acidic Most Acidic X Y >> H X Y > H X Y H - X Y X Y X Y Using the Relative Effect Assumption (As illustrated below.), we can conclude that energies of the conjugate bases increase in the order of those with highest energy electrons on atoms involved in a triple bond, double bond and single bonds. H H C H C: R-CH3 R-CH2 C C: Base H Base G Base GO Acid Rxn. Coord. GN G Acid Rxn. Coord. GC G Acid Rxn. Coord. 2. Suggest structural and/or theoretical arguments to explain the effects observed above. Explanation: The structural differences in the molecules relate to the bond orders of the bonds between the atom holding the newly generated lone pair e-‘s of the conjugate base (the highest energy e-‘s) and an adjacent heavy atom. To explain the energy differences among differently bonded species, we must have a theory that describes bonding in these molecules. So far we have been using a simple Lewis Dot Structure picture of molecular bonding. It has been useful for considering interactions among electron pairs on an atom or electrons with nuclei, but in this case, descriptions of different electron environments on differently bonded atoms, require a more sophisticated approach to molecular structure. One relatively simple bonding theory that was introduced in General Chemistry is Valence Bond theory in which bonds are described as overlap of atomic orbitals and the differing geometries around heavy atoms are ascribed to differences in mixing of its atomic orbitals, hybridization, before bonds are formed between atoms. (See the CH 211 Review of General Chemistry and Mathematics #'s 29. and 30.) Let's use the Valence Bond approach to describe the environments of the electrons in the molecules below. The hybridization of the orbital on the carbon atom, that is used to form the bond to the acidic proton of the conjugate acid and to which the lone pair electrons of the conjugate base is assigned, changes from sp to sp 2 to sp3 as the number of sigma-bonds around the carbon atom increases from 2 to 3 to 4 respectively. The hybridization changes result in different proportions of s and p Acid-Base Rxns & Electron Energy-4 3 character in the hybrid orbital. So there is more s character in the hybrid orbital on the multiply bonded carbon atoms than in the singly bonded atoms. e.g. H C C H H C - C sp orbital sp - hybridized H H H C - C C H H sp2 orbital C H H sp2 - hybridized H H C H - H C sp3 orbital H H H H C C H H H sp3 - hybridized Electron distributions in s orbitals provide more effective interaction between their e-'s and the nucleus (effective nuclear charge) than do p orbitals in the same principle energy level. Consequently, as the s-character of a hybrid orbital increases, the effective nuclear charge felt by the e-'s in the orbital will increase. Thus, the energy of the e-'s will be lowered by increased attractive force as s-character of their orbital increases. This effect is similar to the effect seen when the number of protons in the nucleus increases from C->N->O. Again, the decrease in energy of the e-'s is greater for the higher energy non-bonding electrons (lone pair e-'s in the conjugate base) than for the lower energy bonding electrons in the conjugate acid. Thus, increasing the s-character of the hybrid orbitals on the atom holding the proton lost, lowers the energy of the base form more than it does the acid form. So, increased s-character decreases G thus increasing the acidity of the acid. HC CH CH2 CH2 CH3 CH3 Base Base Base G Gsp Acid Gsp2 G Acid Acid Rxn. Coord. sp pKa G Energy of nonbonding electrons of the conjugate base Acidity of R-H Gsp3 G Rxn. Coord. sp2 Increase Increase Increases Increase Rxn. Coord. sp3 4 Acid-Base Rxns & Electron Energy-4 NOTE: Magnitude of the Effect. There is a much larger difference between sp (50% s) & sp2 (33.3% s) atoms than between sp2 (33.3% s) & sp3 (25% s) atoms. The energy differences parallel the relative changes in % s-character of the orbitals. The overall pKa change from sp to sp3 for otherwise identical compounds is ~20 pK units. The change from sp to sp2 produces a change of ~15 pKa units, while the sp2 to sp3 yields a change of ~5 pKa units.