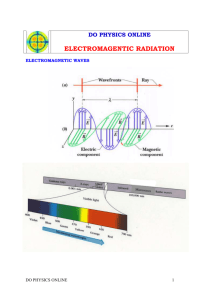
Laser report - WordPress.com
advertisement

PHYS 2053: Homework IX First Draft Justin Courtright Thomas Riley Jessica Belzer 1. Laser: Pumping a two state system a. The emissions appear to be spontaneous, but “stimulated” by the incident light (the emissions change with change in wavelength). There does not seem to be much of a pattern (in time) in the emission. The time during which the electron is in an excited state (n=2) varies, and only one electron moves between the two levels. b. The emitted photons become “fewer and farther between” when the intensity of the light source is changed to a very low level. Otherwise, these photons demonstrate the same characteristics as in the state used for question a. c. The photon emissions occur much more rapidly when the intensity of the light source is changed to a very high level. Excitation and “decay” both happen at a much higher rate than previously. Other than this factor, the same characteristics are present (i.e. one electron moves between the levels, no real pattern in time of the emissions, and the electron moves to the n=2 excited state). d. When the intensity is at a medium level and the lifetime is decreased to 50%, photons are emitted visibly (as opposed to previously when they were not visible within the simulation). These visible emissions are scattered in a random direction, and do not occur each time the electron “decays” down from the n=2 state. Note: even though the previous simulation states did not show visible emissions, the movement of the electron between the energy levels was observed on the right side of the simulation experiment. e. When the intensity is at a medium level, but the lifetime is at 100%, no visible photons are emitted, but all other aspects appear the same (i.e. one electron moves between the levels, no real pattern in time of the emissions, and the electron moves to the n=2 excited state). f. When the light is tuned to a color that has a lower energy (“dark red”) than the preset (red) color, the following is observed. The electron of the atom is not being excited to a higher (n=2) state. Instead, it just remains in the ground state. The distance between the “potential” movement (the distance between the n=1 and n=2 states) of the electron within the shell becomes shorter. g. When the light is tuned to a color that has a higher energy (“dark blue”) than the preset (red) color, the same occurrences as in part f are observed, with the exception that the distance between the “potential” movement (the distance between the n=1 and n=2 states) of the electron within the shell becomes longer. h. Summary: In this part of the simulation, we discovered that visible photons are not always emitted to reflect the movement of the electron between the excited state and ground state. The number of photons emitted is directly correlated to the intensity of the incident light. Lastly, the lifetime of the electron shells affects the “decay time” of the electrons within the excited state, and the visibility of the photon within the simulation. 2. Laser: Pumping a three state system a. When the change is made to a three state system, electrons can be pumped to a higher energy level with yellow, green and blue light in addition to red light. Note: Perhaps these lights may all be distilled to just green light (as this is a fairly common laser and the other light colors surround green), but further research on the subject is needed to draw a conclusion. b. The characteristics observed about the emission events on a per-color basis are as follows: Red All emissions are red (one color) Many of the emitted electrons are given off in pairs Photons are scattered at random Many electrons are at an elevated state (n=2) at a time, and multiple electrons move between the two levels at any given time Yellow All emissions are yellow (one color) About 50% of the emitted electrons are given off in pairs Photons are scattered at random Photons are emitted less often (visibly) in comparison to red light Many electrons are at an elevated state at any time, and multiple move between the levels at any given time Green All emissions are green (one color) Many fewer emissions occur (in comparison to the red light case) Many electrons are at an elevated state (n=2) at a time, and multiple move between the levels at any given time Blue All emissions are blue (one color) Very few visible photons are emitted Photons are scattered at random, and often they are released in pairs Many electrons are at an elevated (n=2) state at a time, and multiple move between the levels at any given time Note: all of the photons seem to be spontaneously emitted (for visible emissions), but are “stimulated” to an excited state by the incident light source. Also, cases where a photon stimulates an atom to emit a second photon are seen as described where the atom releases pairs of photons. c. When the “display photons emitted from upper state” option is selected, these photons represent the transition from the n=3 to the n=1 state. Photons from 31 are green light, while photons from 21 are red. The region of the spectrum to which they belong is visible light. d. When the lifetime for the n=3 level is increased, photons are emitted less often, but in greater bursts. This is due to the resulting slowed decay from the n=3 level. e. When the lifetime for the n=2 level is increased, photons are emitted less often. The number of electrons in this level increases as directly correlated with lifetime. This is because the decay from this level is slowed. The most electrons of the 3 levels are in the n=2 level. (The n=3 level had a lifetime of 50%). f. Summary: Increased lifetime at a given level causes a slowed decay from that level. The visible emissions appear spontaneous in nature, but cases where a photon stimulates an atom to emit a second photon are seen as described where the atom releases pairs of photons. We also noted that generally as the wavelength of light decreases, photons are emitted less often. 3. Making a laser with a three state system a. Conditions tha make a good laser include: i. The use of mirrors, with a reflectivity of about 98% (90+%, but less than 100%) ii. A mid-length lifetime for a “medium” state (ex. N=2 in a 3 state system) iii. The use of red or green light b. Note: I used green light in my observations 4. A small helium-neon laser produces a light beam with an average power of 3.5mW and a diameter of 2.4 mm. a. Note: Please see attached calculations b. “The 633 nm line was found to have the highest gain in the visible spectrum, making this the wavelength of choice for most HeNe lasers.” – Source: Wikipedia: Helium-Neon Laser http://en.wikipedia.org/wiki/Helium-neon_laser (Note: this information was cross referenced with other sources)