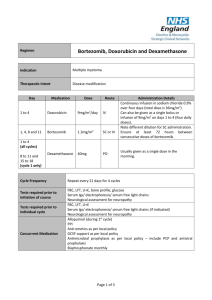
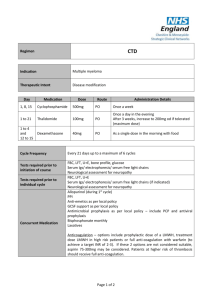
1, 8, 15
advertisement
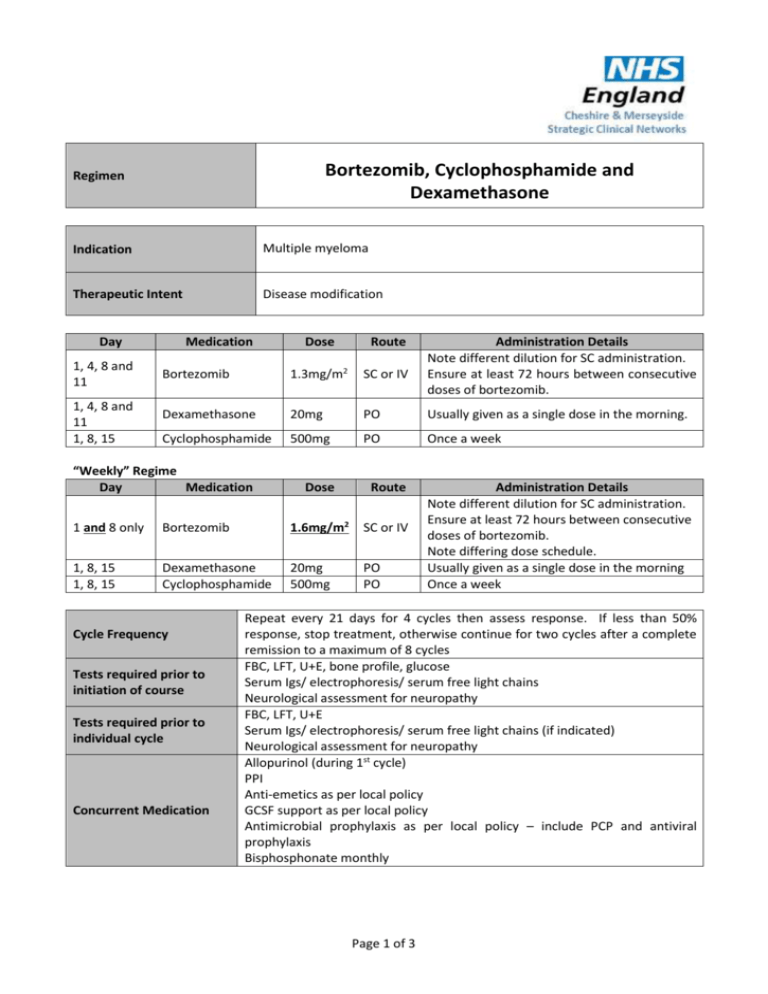
Bortezomib, Cyclophosphamide and Dexamethasone Regimen Indication Multiple myeloma Therapeutic Intent Disease modification Day 1, 4, 8 and 11 1, 4, 8 and 11 1, 8, 15 Medication Dose Route Bortezomib 1.3mg/m2 SC or IV Dexamethasone 20mg PO Usually given as a single dose in the morning. Cyclophosphamide 500mg PO Once a week “Weekly” Regime Day Medication Dose Route 1 and 8 only Bortezomib 1.6mg/m2 SC or IV 1, 8, 15 1, 8, 15 Dexamethasone Cyclophosphamide 20mg 500mg PO PO Cycle Frequency Tests required prior to initiation of course Tests required prior to individual cycle Concurrent Medication Administration Details Note different dilution for SC administration. Ensure at least 72 hours between consecutive doses of bortezomib. Administration Details Note different dilution for SC administration. Ensure at least 72 hours between consecutive doses of bortezomib. Note differing dose schedule. Usually given as a single dose in the morning Once a week Repeat every 21 days for 4 cycles then assess response. If less than 50% response, stop treatment, otherwise continue for two cycles after a complete remission to a maximum of 8 cycles FBC, LFT, U+E, bone profile, glucose Serum Igs/ electrophoresis/ serum free light chains Neurological assessment for neuropathy FBC, LFT, U+E Serum Igs/ electrophoresis/ serum free light chains (if indicated) Neurological assessment for neuropathy Allopurinol (during 1st cycle) PPI Anti-emetics as per local policy GCSF support as per local policy Antimicrobial prophylaxis as per local policy – include PCP and antiviral prophylaxis Bisphosphonate monthly Page 1 of 3 Dose Modifications Hepatic Renal Patients with mild hepatic impairment do not require a dose adjustment and should be treated per the recommended dose. Patients with moderate or severe hepatic impairment should be started on bortezomib at a reduced dose of 0.7 mg/m2 per injection during the first treatment cycle, and a subsequent dose escalation to 1.0 mg/m2 or further dose reduction to 0.5 mg/m2 may be considered based on patient tolerability. (See SPC for further details). Serum Creatinine (micromol/L) Modification >300 Omit cyclophosphamide The pharmacokinetics of bortezomib is not influenced in patients with mild to moderate renal impairment therefore, dose adjustments are not necessary for these patients. It is unknown if the pharmacokinetics of bortezomib are influenced in patients with severe renal impairment not undergoing dialysis. Since dialysis may reduce bortezomib concentrations, bortezomib should be administered after the dialysis procedure. Haematological Platelets (x 109/L) Neutrophils (x 109/L) ≥ 1 x 109/L Modification and ≥ 50 x 109/L 100% dose. 0.75 - 1 x 109/L or 30 - 50 x 109/L Delay cyclophosphamide 1 week (continue dexamethasone). Restart at same dose when neutrophils and platelets recovered as above. If recurrent, i.e. if neutrophils 9 <1.0x10 /L and platelets < 50x109/L on day 1 of subsequent cycles (when previously > than these levels), delay cyclophosphamide and consider dose reduction of cyclophosphamide. If the patient was receiving 500 mg weekly, reduce to 400 mg, if 400 mg reduce to 300 mg, if 300 mg reduce to 200 mg. Consider adding G-CSF for 2 to 3 days per cycle or week (usually only requires low doses). Page 2 of 3 ≤ 0.75 x 109/L Peripheral neuropathy or ≤ 30 x 109/L Severity of neuropathy Grade 1 (paraesthesia, weakness and/or loss of reflexes) with no pain or loss of function Bortezomib should be withheld with these blood counts on a bortezomib dosing day (other than day 1). If several bortezomib doses in a cycle are withheld ( ≥ 3 doses during twice weekly administration) the dose should be reduced by 1 dose level from 1.3 mg/m2 to 1 mg/m2, or from 1 mg/m2 to 0.7 mg/m2). Withhold cyclophosphamide dose until count recovers and consider starting at a reduced dose. Modification None. Grade 1 with pain or Grade 2 (interfering with function but not with activities of daily living) Reduce bortezomib to 1.0 mg/m2 Grade 2 with pain or Grade 3 (interfering with activities of daily living) Withhold bortezomib treatment until symptoms of toxicity have resolved. When toxicity resolves reinitiate bortezomib treatment and reduce dose to 0.7 mg/m2 and change treatment schedule to once per week. Grade 4 (sensory neuropathy which is disabling or motor neuropathy that is life threatening or leads to paralysis) and/or severe autonomic neuropathy Discontinue bortezomib. Additional Information The current NICE recommendation is to give 4 cycles initially, then review and proceed as follows: If responding (defined as 50% of greater reduction in paraprotein i.e. CR or PR) continue until 2 cycles after CR to a maximum of 8 cycles. For non-responders after 4 cycles, discontinue treatment. The manufacturer will be required to reimburse the full cost of bortezomib under the Velcade® Response Scheme. References http://emc.medicines.org.uk NICE technology appraisal guidance 228 July 2011. Author Pharmacy CNG Approved & Checked by Haematology CNG (Review Date = Sept 2017) Page 3 of 3