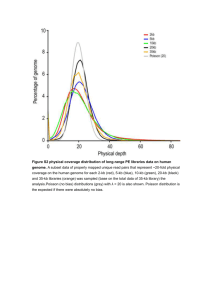
The Primary Cilia Dyskinesia Gene HEATR2 Plays
advertisement

The Primary Cilia Dyskinesia Gene HEATR2 Plays a Conserved Role in Assembly of Ciliary Motile Apparatus Supplementary Materials and Methods Christine P. Diggle1,9, Daniel J. Moore2,9, Girish Mali3,9, Petra zur Lage2, Aouatef AitLounis4, Miriam Schmidts5, Amelia Shoemark6, Amaya Garcia Munoz7, Mihail R. Halachev3, Philippe Gautier3, Patricia Yeyati3, David T. Bonthron1, Ian M. Carr1, Bruce Hayward1, Alexander F. Markham1, Jilly E. Hope2, Alex von Kriegsheim7, Hannah M. Mitchison5, Ian J. Jackson3, Bénédicte Durand8, Walter Reith4, Eamonn Sheridan1,10, Andrew P. Jarman2,10 and Pleasantine Mill3,10. 1. School of Medicine, University of Leeds, Leeds, United Kingdom. 2. Centre for Integrative Physiology, School of Biomedical Sciences, University of Edinburgh, Edinburgh, United Kingdom. 3. MRC Human Genetics Unit, Institute of Genetics and Molecular Medicine at The University of Edinburgh, Western General Hospital, Edinburgh, United Kingdom. 4. Department of Pathology and Immunology, Faculty of Medicine, Université de Genève, Geneva, Switzerland. 5. Molecular Medicine Unit and Birth Defect Research Center, Institute of Child Health, University College London, London, United Kingdom. 6. Paediatric Respiratory Department, Royal Brompton Hospital, London, United Kingdom. 7. Systems Biology Ireland, University College Dublin, Belfield, Dublin, Ireland. 8. Centre de Génétique et de Physiologie Moléculaire et Cellulaire, UMR 5534 CNRS, Université Claude Bernard Lyon 1, Villeurbanne, France. 9. These authors contributed equally to this work. 10. These authors jointly supervised this work. Correspondence: pleasantine.mill@igmm.ed.ac.uk, andrew.jarman@ed.ac.uk, e.sheridan@leeds.ac.uk Running head: HEATR2 is required for Cilia Motility. Ribonuclease protection assay (RPA) Poly A+ RNA was isolated using the poly(A)purist kit (Ambion). To generate riboprobes, RTPCR products were generated from control and PCD patient cDNA using the Phusion DNA polymerase (NEB), and RPA primers (Table S4). Additional 3’ A’s were added prior to cloning in pGEMTeasy (Promega). Biotinylated riboprobes were produced using the Megascript kit (Ambion), and hybridisation of polyA selected RNA and probes, and subsequent digestion was carried out using the RPAIII kit according to the manufacturer’s instructions (Ambion). Samples were separated on a 6% denaturing acrylamide gel, blotted onto a nylon membrane and bands detected using the BrightStar system (Ambion). Band intensity was quantified using a Biorad Imager. The size of the bands was taken into account when determining the level of transcipt. HEATR2 orthologue identification and axonemal dynein studies Potential orthologous sequences were collected using one of the following methods: direct use of existing annotation in Ensembl (Release 68 (July 2012), Flicek et al, 2012) and Ensembl Genomes (Release 15 (August 2012), Kirsey et al, 2012), blastp and tblastn searches (Altschul et al, 1997) using the relevant species database (NCBI http://www.ncbi.nlm.nih.gov/protein, JGI genome portal http://genome.jgi-psf.org/ (Grigoriev et al, 2012), the phytozome database http://www.phytozome.net/ (Goodstein et al, 2012). Orthology was tested with a reciprocal blast search of the candidate protein against the human proteome, and reciprocal best hits were labeled as orthologous sequences. In some cases, the reciprocal hit was second to best, but this can be explained in cases were 1 ancestral DNAH gene has been duplicated in vertebrates (for example, DNAH3 and 7). In such cases, we decided to label the candidate as an orthologue. Whenever a gene prediction was absent or incomplete, we also made our own protein prediction using genewise (version wise2-1-23c, Birney et al, 2004). All HEATR2 sequences were aligned using Clustalw (version 2.0.12 , Higgins et al, 1996). Percentage of identities were calculated from the clustal multiple alignment using Genedoc (version 2.7.000, Nicholas et al, 1997). Conservation of Fox and X-box binding motifs in HEATR2 promoter studies Sequences used for these studies were taken from Ensembl release 64, September 2011: Human GRCh37 assembly of the human genome [February 2009]. We analysed position Ch7:771338 to the gene start (Ch7:766338) and the syntenic region in other species. We used dna-pattern, a program from the RSAT (Regulatory Sequence Analysis Tools) package (van Helden, 2003), with the above consensus sequence. We also used the Vista server (http://genome.lbl.gov/vista/index.shtml) and Pipmaker (http://pipmaker.bx.psu.edu/pipmaker/) to align these regions and detect conserved sequences. Using the human sequence available through the ENCODE project, we also analyzed a combination of epigenetic markers including Histones markers, CTCF and Pol2 markers detected by ChIP-seq, on several cell lines whereby the raw data has applied multivariate Hidden Markov models to 9 factors in 9 cell types (see http://genome.ucsc.edu/cgibin/hgc?hgsid=246057575&o=50384596&t=50384996&g=wgEncodeBroadHmmGm12878HM M) to assign the most likely state for each 200bp interval (i.e. strong promoter, weak promoter, strong enhancer, etc). This was a powerful complementary source of information to judge the conserved regions covered by our potential Fox and X-box binding sites. Heatr2 localization studies NIH-3T3 cells were transiently co-transfected with full length mouse Heatr2 with a C-terminal turboGFP tag (Origene) and mouse Arl13b with a C-terminal mKate2 fusion (gift from Dr. E. Hall). DNA plasmids using Neon transfection system (Invitrogen) according to manufacturer’s instructions. Briefly, cells were resuspended in Solution R and mixed with 0.5μg of each plasmid (total 1µg DNA) and the mixture was taken up in 10µL Neon Tips for microporation. 3 transfection reactions were plated into each well of a glass-bottomed 24 well plate (Iwaki) in prewarmed medium without antibiotics. After 24 hours, cells were serum starved, and imaging began after 24h serum starvation. One hour prior to live imaging, the media was changed to prewarmed phenol red-free DMEM-gfp (Evrogen) substitute with 4mM L-glutamine, 1% penicillin/streptomycin. Live imaging was then performed in a heated incubation chamber using a Nikon A1R confocal microscope and time-lapses processed using Nikon NIS-Elements 4. Nasal biopsy immunofluorescence protocol Protocol: Respiratory epithelial cells were obtained by nasal-brush biopsy and suspended in cell-culture medium. Samples were spread onto glass slides, air dried, and stored at –80°C until use. For axonemal dynein, slides were defrosted, fixed in 4% paraformaldehyde (PFA) for 3 minutes, washed 5 times in PBS, incubated in 0.05% TritonX-100 for 3 minutes, washed 5 times, blocked in 5% BSA for 1 hour at RT followed by incubation with primary antibody at 4°C O/N, washed 5 times in PBS, incubated for 2 hours in secondary antibody at RT, washed 5 times, mounted in Vectashield (Vector Labs) and imaged using a Zeiss LSM710 confocal microscope. Pictures were processed using ImageJ software. For endogenous HEATR2 staining, slides were defrosted, either fixed 10 minutes in ice-cold methanol at -20°C then briefly air-dried or 4% PFA/PHEM (120mM PIPES, 140mM HEPES, 20mM EGTA, 16mM MgSO4, pH7) for 10 minutes at 37°C. then rinsed in TBST (0.05% TritonX-100) repeatedly, blocked in 5% donkey serum for 60 minutes RT, followed by incubation with primary antibodies for 60 minutes RT or 4°C O/N diluted in 1% donkey serum, washed in TBST repeatedly, incubated for 60 minutes RT with secondary antibodies and DAPI in 1% donkey serum, washed in TBST repeatedly and mounted in Prolong Gold (Molecular Probes) according to manufacturer’s protocol. Images were captured using a Nikon A1R confocal microscope and processed using Nikon NIS-Elements 4. Supplemental Bibliography Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ. 1997. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Research 25: 3389 402. Birney E, Clamp M and Durbin R. 2004. GeneWise and Genomewise. Genome Research 14: 988-995, 2004 Flicek P, Amode MR, Barrell D, Beal K, Brent S, Carvalho-Silva D, Clapham P, Coates G, Fairley S, Fitzgerald S, Gil L, Gordon L, Hendrix M, Hourlier T, Johnson N, Kähäri AK, Keefe D, Keenan S, Kinsella R, Komorowska M, Koscielny G, Kulesha E, Larsson P, Longden I, McLaren W, Muffato M, Overduin B, Pignatelli M, Pritchard B, Riat HS, Ritchie GR, Ruffier M, Schuster M, Sobral D, Tang YA, Taylor K, Trevanion S, Vandrovcova J, White S, Wilson M, Wilder SP, Aken BL, Birney E, Cunningham F, Dunham I, Durbin R, Fernández-Suarez XM, Harrow J, Herrero J, Hubbard TJ, Parker A, Proctor G, Spudich G, Vogel J, Yates A, Zadissa A, Searle SM. 2012. Ensembl 2012. Nucleic Acids Research 2012 40 Database issue:D84-D90 Goodstein DM, Shu S, Howson R, Neupane R, Hayes RD, Fazo J, Mitros T, Dirks W, https://plus.google.com/u/0/meHellsten U, Putnam N, Rokhsar DS. 2012. Phytozome: a comparative platform for green plant genomics. Nucleic Acids Res. 2012 Jan;40(Database issue):D1178-86. Epub 2011 Nov 22. Grigoriev IV, Nordberg H, Shabalov I, Aerts A, Cantor M, Goodstein D, Kuo A, Minovitsky S, Nikitin R, Ohm RA, Otillar R, Poliakov A, Ratnere I, Riley R, Smirnova T, Rokhsar D, Dubchak I. 2012. The genome portal of the Department of Energy Joint Genome Institute. Nucleic Acids Res. 2012 Jan;40(Database issue):D26-32. Epub 2011 Nov 22. Higgins, DG, Thompson, JD and Gibson, TJ. 1996. Using CLUSTALW for multiple sequence alignments. Methods in Enzymology 266: 383-402. Kersey PJ, Staines DM, Lawson D, Kulesha E, Derwent P, Humphrey JC, Hughes DS, Keenan S, Kerhornou A, Koscielny G, Langridge N, McDowall MD, Megy K, Maheswari U, Nuhn M, Paulini M, Pedro H, Toneva I, Wilson D, Yates A, Birney E. 2012. Ensembl Genomes: an integrative resource for genome-scale data from non-vertebrate species. Nucleic Acids Res. 2012 Jan;40(Database issue):D91-7. Epub 2011 Nov 8. Nicholas, K.B., Nicholas H.B. Jr., and Deerfield, D.W. II. 1997 GeneDoc: Analysis and Visualization of Genetic Variation, EMBNEW.NEWS 4:14