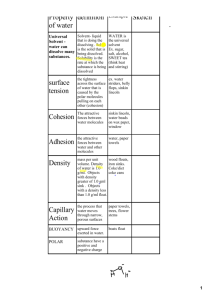
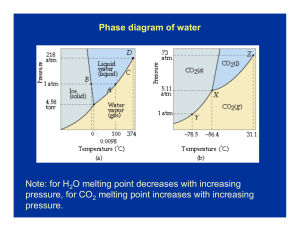
Solutions_answers
advertisement

Solutions Answer Sheet Chapter 14 14) (a) No, (b) Yes, it is. Because increasing the pressure does not significantly increase the contact of the solutes with the solvent. 23) For dilute aqueous solutions near room temperature. Molarity is most useful when using equipment that measures liquids by volume since molarity is based on volume. 36) Two possible explanations: (1) Solute molecules reduce the number of solvent molecules at the surface of the solvent, or (2) Solute molecules take energy from solvent molecules at the surface. 91) Colloidal dispersions are made from large aggregations of surfactant molecules to form colloids and the colloids are dispersed in the solvent. A true solution is made from solute molecules dispersed individually in the solvent.