Discuss
advertisement

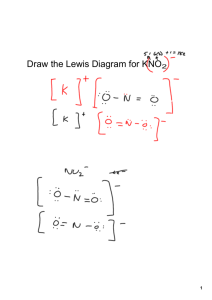
B9 & B.10 Supplement: Ions and Ionic Compounds. 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. Cation Ca2+ Na+ NH4+ CU2+ Fe3+ K+ Na+ Ag+ Zn2+ Ba2+ Al3+ K+ Mg2+ Ba2+ Zn2+ Ca2+ Fe3+ Pb2+ Pb2+ Li+ Anion O2ClNO3OH- Formula CaO NaCl NH4NO3 Cu(OH)2 Name Calcium oxide Sodium chloride Ammonium nitrate Copper(II) hydroxide SO42SO32PO43S2BrF1PO43S2O2SO42PO43HCO3ClCO32BrNO3- Fe2(SO4)3 K2SO3 Na3PO4 Ag2S ZnBr2 BaF2 AlPO4 K2S MgO BaSO4 Zn3(PO4)2 Ca(HCO3)2 FeCl3 PbCO3 PbBr2 LiNO3 Iron(III) sulfate Potassium sulfite Sodium phosphate Silver sulfide Zinc bromide Barium fluoride Aluminum phosphate Potassium sulfide Magnesium oxide Barium sulfate Zinc phosphate Calcium hydrogen carbonate Iron(III) chloride Lead(II) carbonate Lead(II) bromide Lithium nitrate Section B Supplement 1. A characteristic exhibited by a substance that does not involve interacting with or changing into other substances. 2. Several answers are possible: odor, density, boiling point, freezing point, etc. 3. The mixture is a colloid. There are no particles large enough to settle out (ruling out the possibility of a suspension), but small particles ARE present―as light was scattered. Therefore, it is not a solution (which would contain no particles) and IS a colloid. 4. Draw a model of a solution in which water (H2O) is the solvent and carbon dioxide gas (CO2) is the solute. Student models should show a mixture of water molecules and carbon dioxide molecules. There should be noticeable more water molecules than carbon dioxide molecules. 5. a. NaCl compound b. C element c. CO compound d. KNO3 compound 6. Draw a model of a molecular compound made up of two atoms of B and two atoms of E. Student answers may include variations of: = B and = E 7. Complete the following table by naming the elements present and the number of each type of atom. Compound Element Names Number of Atoms of Each Element H2SO4 H = Hydrogen; S = Sulfur; O = Oxygen H2 S1 O4 CO2 C = Carbon; O = Oxygen C1 O2 CaCl2 Ca = Calcium; Cl = Chlorine Ca 1 Cl 2 Al2S3 Al = Aluminum; S = Sulfur Al 2 S 3 NH4NO3 N = Nitrogen; H = Hydrogen; O = Oxygen N2 H4 O3 8. N2 + 3H2 → 2NH3 9. Protons are positive and electrons are negative. They are equal and opposite in change. 10. Element Protons Electrons Neutral? Gold 79 76 No, positive Helium 2 2 Yes Fluorine 9 10 No, negative. Sodium 11 11 Yes. 11. a. K+ cation b. Ag neutral 12. a. hydrogen with one proton and one electron H b. lithium with three protons and two electrons Li1+ c. fluorine with nine protons and ten electrons F113. Cation Anion Formula c. N3- anion d. Ba2+ cation Name Mg2+ O2- MgO magnesium oxide Al3+ Cl- AlCl3 aluminum chloride Mg2+ NO3- Mg(NO3)2 magnesium nitrate NH4+ CO32- NH4(CO3)2 ammonium carbonate 14. a. A precipitate is an insoluble substance that separates from water (as a solid) in solution. b. Chloride ion is present in Stream A. c. It is not possible to conclude whether or not chloride is present in Stream B. The best we can say is that chloride ion is not present in detectable levels in Stream B.