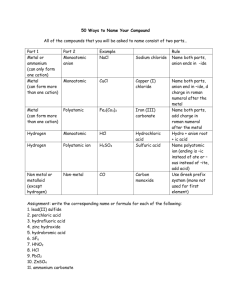
How do balance an ionic Formula formula
advertisement

Equation Practice How do balance an ionic Formula formula? First, note that the name is in two parts ( a cation followed by an anion). These ions will be listed with their name formula and charge on the “valency table” (Which you promise not to lose!) Example Iron (iii) is a cation. You will see that it is Fe3+ Chromate is CrO42- . It is an anion. It is actually made from a group of 5 atoms and has a charge of -2. You must treat the group as one thing. To write a formula for Iron (iii) Chromate: Replace name with symbols from table. Fe3+ CrO42The charges do not balance. Take 2 of the iron and 3 of the chromate to balance the charges as 6+ and 6 – Write it : Fe2 (CrO4)3 :- we used brackets because chromate is made from more than 1 atom. We have to keep it as a group. Your turn! Write balance formula for the following: I. II. III. IV. V. Silver Phosphate Magnesium Chloride. Ammonium Nitride Copper (ii)Sulphite Aluminium carbonate. 1 Equation Practice What is a molecular Formula? Let’s consider the formula for sugar C12H22O11. This means one molecule of sugar. The molecule has 12 atoms of carbon, 12 atoms of Hydrogen, 11 atoms of oxygen. Your turn again. Explain the following formulas. Acetic acid C2H4O2 Benzene C6H6 Carbon tetra chloride C Cl4 Formulas you need to Know. Water is H2O, Carbon Dioxide is CO2, Ammonia s NH3, Hydrogen, oxygen, Chlorine, Fluorine, Nitrogen are diatomic gases. You write the formula with a little 2. See page 52. Acids you need to know. Sulphuric acid H2SO4 cation is H+ anion is SO42- Hydrochloric acid HCl cation is H+ anion is Cl- Nitric acid H NO3 cation is H+ anion is NO3- Acetic (ethanoic acid) CH3COOH cation is H+ anion is CH3COO- 2 Equation Practice How do we balance double displacement reactions? Not the reactants are ionic compounds. The name is two parts. cation anion. Write the names of the two reactants in words with a + sign between. Then swap anions to write products in words after the arrow. Example Sodium chloride solution is mixed with silver nitrate solution. Step 1 word equation calcium chloride + silver nitrate calcium nitrate + silver chloride. Step 2 use the valence tables to write the symbols. Ca2+ Cl - + Ag +NO3- Ca2+ NO3- + Ag+ Cl- Step 3 balance individual formulas using charges. Ca Cl2 + Ag NO3 Ca (NO3)2 + Ag Cl Ca (NO3)2 + 2Ag Cl Step 4 use coefficients to balance overall Ca Cl2 + 2Ag NO3 Step 5 add states to show precipitates Ca Cl2(aq) + 2Ag NO3(aq) Ca (NO3)2(aq) + 2Ag Cl(aq) Try these Copper nitrate solution mixed with sodium carbonate solution Potassium phosphate solution mixed with lead(ii) chloride solution Iron (iii) nitrate solution mixed with sodium hydroxide solution Barium nitrate solution mixed with sodium sulphate solution Calcium chloride solution mixed with ammonium carbonate solution 3 Equation Practice Reactions with acids The acid is like a salt where the cation is H+ If it reacts with a metal the products are salt and Hydrogen gas. If it reacts with a carbonate or hydrogen carbonate then products are salt + carbon dioxide + water. If it reacts with metal oxide or metal hydroxide then products are salt + water. Try these Hydrochloric acid + magnesium metal Nitric acid + copper carbonate Sulphuric acid + lead (ii) oxide Acetic acid + sodium Hydrogen carbonate. Nitric acid + Zinc metal Hydrochloric acid + calcium Hydroxide. Single Displacement Metal 1 + Metal 2 salt metal 2 + metal 1 salt Try these Copper metal + silver nitrate Iron metal + copper chloride Magnesium metal + nickel nitrate. Combustion practice Write balances reactions for the following burnt in air. Ethanol (C2H6O), Pentane (C5H12), propanol (C3H8O) 4