Titration and Neutralization Lab Report: HCl & NaOH
advertisement

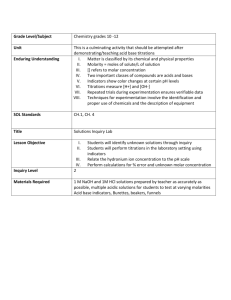
LAB ON TITRATION AND NUETROLIZATION MADE BY BENAZIR AYAMY TEACHER MR. VINCET CLASS GRADE 12 COLLEGE PREPRATION INTRODUCTION The acid-base titration is the determiantion of concentration of an acid or base by nuetulizing the acid/base with an acid or base of unkown concentration. Titration is the volumetric chemical analysis to determine the concentration of a substance in solution.neutrulization is a reaction to nuetrolize the acid with base or the base with acid. In this experiment we are using HCL to nuetrolize NAOH and indicator phenolphthalein, that change the colour the point that the colour change is called endpoint. MATERIAL Apparatus: 1. volumetric flask 2. 10mL measuring cylinder 3. Pipette 4. 50mL burette 5. Beaker 6. Erlenmeyer flask 7. Retord stand 8. White tile 9. Stopwatch 10. Pipette bulb Chemicals: 1 NAOH solution 2. 0.09M HCL solution 3. Distilled water 4. Phenolphthalein PROCEDURE GIVEN IN ABOVE OBSERVATION READING VOLUME OF HCL V1 FINAL BURETTE READING VOLUME OF HCL V2 VOLUME OF ACID CONSUMED (ml) TITRANT V= V2-V1 10ml 0 12.3 V=12.3-0=12.3 2 10ml 0 11.9 V=11.9-0=11.9 3 10ml 0 11.8 V=11.8-0=11.8 TRIAL NUMBER VOLUME OF ALKALI (ml) INITIAL BURETTE 1 While doing this experiment when we add phenolphthalein to the NAOH the color of base change to pink but by adding HCL the color change slightly to bright pink at the end by adding one drop of HCL the color of NAOH totally change to white so we saw that how HCL neutralize the NAOH. CALCULATION CIVI=CFVF CI*10ml=0.09M*12ml CI= 0.09M*12ml/10ml CI=0.108M CI Represent the no of concentration of NAOH or the Morality (0.108M) C=n/v n=c*v n=0.108M*12ml n=1.296M/ml MmNAOH= (22.98+16+1.01) MmNAOH= 40.01gr/mol (the Molor mass of NAOH) n=m/Mm m=n*Mm m=1.296M/ml*40.01gr/mol m=51.9gr (the mass of NAOH present in 1Liter) CONCLUSION In conclusion we can say or we find that how does the titration and neutralization happen and how and acid neutralize the base. DISCUSSION TITRANT during titration the solution in burette called the TITRANT. The three indicators that we can use are (methyl orange, bramothymol blue and phenolphthalein). End point is the point when the color of base was change at the end by adding one drop acid to the base the color of base totally changes to natural. By washing Erlenmeyer flask yes it will effect to the concentration of NAOH. SOURCE OF EXPERIMENT ERROR The error can be while we are pipetting the acid or the base we didn’t do it correctly. In titration we forgot to do the titration of pipette before taking the acid or the base. The biker that we are using is not properly dry and while adding acid to the base we were not doing that properly because it need more patience we have add a very little amount each time and at the end when the color is slightly bright so we have to be careful to the last drop.