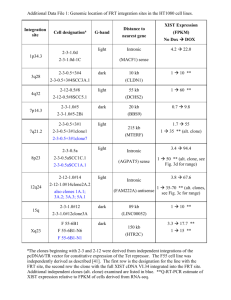
C. elegans epistasis You are interested in studying vulval
advertisement

C. elegans epistasis You are interested in studying vulval development in C. elegans! Because you really truly enjoy examining the bag-of-worms phenotype that results when eggs hatch inside their mother, and the creepiness of multi-vulval worms delights you! Yup, pretty cool, right? You have the genotypes listed on the following table. gf = over-expression (you have a line with a transgene containing multiple copies of each gene) lf= complete loss of function) Single mutants Multivulval lin-15(lf) lin-3(gf) let-60(gf) lin-1(lf) lin-3(lf) let-23(lf) let-60(lf) lin-45(lf) Vulvaless lin-3(lf) let-23(lf) let-60(lf) lin-45(lf) lin-15(lf) Multivulval Vulvaless Vulvaless Vulvaless lin-3(gf) Multivulval vulvaless No data No data let-60(gf) Multivulval Multivulval Multivulval Vulvaless lin-1(lf) Multivulval Multivulval Multivulval Multivulval A) Which genes in this pathway are positive regulators of vulval formation? Which genes are negative regulators of vulval formation? Negative regulators: lin-15, lin-3, lin-1, lin-45 (lin-45 is tricky because it’s involved in double inhibition), Positive regulators: let-60, let-23 B) How would you order these genes in a pathway? Lin-3 --| lin-15 --| let-23 let-60 lin-45 --| lin-1 --| vulval formation C) To complicate matters futher – another lab has recently identified another mutant, glp-1 which lacks a germline altogether. glp-1 mutants also don’t have a normal vulva. Can you order glp-1 in your pathway? Purely from an epistatis pathway, yes. glp-1 since it ablates the germline altogether is technically epistatic to all the lin- genes. However, in reality because glp-1 specifies cells that are precursors to normal germline development, glp-1; lin-15 double mutants don’t really tell you much about the lin-15 pathway. This is similar to the original example Jeff used when introducing epistatis (ie in a hairless mouse, talking about which gene specifies coat color is not particularly informative) Each cell in the Drosophila wing produces a hair that points towards the tip of the wing. Researchers wondered how each cell knew which way the wing tip was. Screens were carried out and mutants identified where the consistent hair orientation is disrupted. You obtain one of the mutant strains which contains a mutation in frizzled and decide to characterize it further. Wildtype PCP mutant Problem 1: You first decide to ask whether frizzled is acting autonomously or non-autonomously to coordinate hair orientation. What technique will you use? Ans: mosaic analysis Problem 2: What is the genotype of the fly that you will analyze? Ans: wing driver: flp/+; FRT fz/FRT tub:GFP Problem 3: How will you obtain this fly? Assume you have three starting strains consisting of 1) engrailed::FLP (on the second chromosome) 2) FRT, fz15 (on the third chromosome) and 3) FRT tubulin:GFP. What fraction of the progeny will be what you want? Cross1 en:FLP x FRT tub:GFP Cross 2 en:FLP/+; FRT tub:GFP/+ x FRT, fz ¼ of progeny will be right can be identified by looking for GFP- clones in the wing. Problem 4: What do you expect to see if frizzled is cell autonomous or non autonomous? Note: you have to look at the edge of the clone to check for autonomy vs non autonomy. Fun fact: fz like all the PCP genes has both autonomous and non autonomous effects. Problem 5: Talking to your lab mates you realize that lots of them are going to be doing the same experiment you just did only using their favorite genes. As a public service you decide to make the engrailed:FLP; FRT tubulin-GFP fly line. How will you go about doing this? For this problem you cannot distinguish by fluorescence one copy vs two of GFP and the engrailed: FLP is unmarked. Explain at every step how you will select the flies you want. Hint: your lab has a L/Cyo; TM3/TM6 double balanced stock. Ans En:FLP x L/Cyo; TM3/TM6 select non lobed progeny En:FLP/Cyo; TM3/+ x L/Cyo; TM3/TM6 select non lobed both TM3 and TM6 progeny En: FLP/Cyo; TM3/TM6 x L/Cyo; FRT tub:GFP/TM3(these flies obtained from basically the same series of crosses only using the FRT line) select non lobed, GFP positive w/ TM6 marker flies En:FLP/Cyo; FRT tub:GFP/TM6 x to itself; select progeny w/o balancers