15.Test Review CW
advertisement

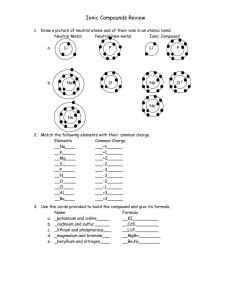
Nomenclature Review 1. What are type I compounds are composed of? 2. Given the compound formula of a type II compound, example FeO, how does one determine the charge on the cation? 3. When working with type III compounds why is charge not considered? 4. What are type II compounds composed of? 5. What does the Roman numeral in a type II compound represent? 6. Why can the quantities of a type I compound be ignored when naming the compound? Example: MgF2 is called Magnesium fluoride, the 2 is ignored. 7. What are type III compounds composed of? Name each of the following: 8. Al2S3 9. FeO 10. SO2 11. MgO 12. MgCl2 13. ZrN 14. CsI 15. Na3N 16. CBr4 17. BeCl2 18. NiCl 19. LiBr 20. KCl 21. MgI2 22. IBr 23. MnO2 24. Why can’t any of the polyatomic ions be part of a type III compound? 25. What type of compound (I, II, or III) does NH 4+ always form and why? Write the compound formula for the following: 26. Iron (III) bromide 27. Calcium iodide 28. Iodine pentafluoride 29. Vanadium (III) oxide 30. Lithium sulfide 31. Magnesium iodide 32. Cobalt (II) Sulfide 33. Manganese (IV) carbonide 34. Oxygen difluoride 35. Potassium Chloride 36. Barium phosphide 37. Chromium (III) nitride 38. Aluminum sulfide 39. Iron forms both 2+ and 3+ cations. Write the formulas for the oxide, sulfide, and chloride compound of each iron cation and give the appropriate name for each compound. 40. Nitrogen and oxygen form numerous binary compounds, including NO, NO2, N2O4, N2O5, N2O. Give the name of each of these compounds. 41. Most metallic elements form oxides, and often the oxide is the most common compound of the element that is found in the earth’s crust. Write the formulas for the oxides of the following metallic elements: a. b. c. d. e. Potassium Magnesium Zinc(II) Lead(II) Aluminum 42. In which of the following pairs is the name incorrect? Give the correct name for the formulas indicated. f. g. h. i. j. Ag2O, disilver monoxide N2O, dinitrogen monoxide Fe2O3, Iron (II) oxide PbO2, lead oxide Cr2O3, chromium (III) oxide