Regents Chemistry 2012-2013
advertisement
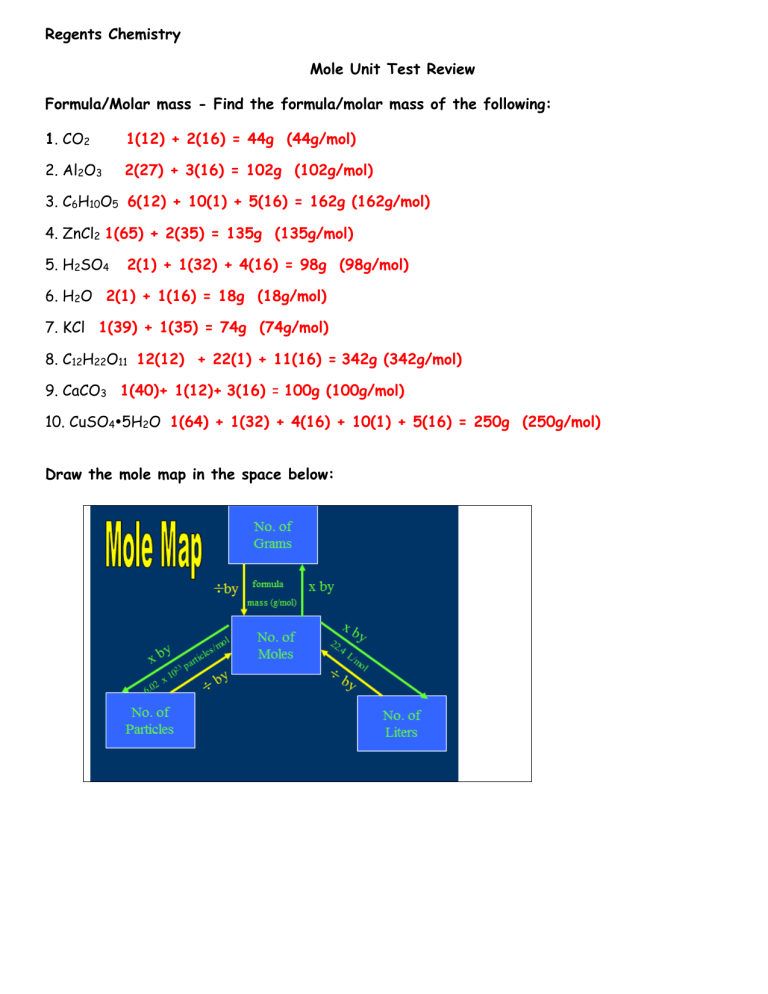
Regents Chemistry Mole Unit Test Review Formula/Molar mass - Find the formula/molar mass of the following: 1. CO2 1(12) + 2(16) = 44g (44g/mol) 2. Al2O3 2(27) + 3(16) = 102g (102g/mol) 3. C6H10O5 6(12) + 10(1) + 5(16) = 162g (162g/mol) 4. ZnCl2 1(65) + 2(35) = 135g (135g/mol) 5. H2SO4 2(1) + 1(32) + 4(16) = 98g (98g/mol) 6. H2O 2(1) + 1(16) = 18g (18g/mol) 7. KCl 1(39) + 1(35) = 74g (74g/mol) 8. C12H22O11 12(12) + 22(1) + 11(16) = 342g (342g/mol) 9. CaCO3 1(40)+ 1(12)+ 3(16) = 100g (100g/mol) 10. CuSO45H2O 1(64) + 1(32) + 4(16) + 10(1) + 5(16) = 250g (250g/mol) Draw the mole map in the space below: Perform the following conversions: 11. 2 moles of NH3 to grams (2moles)(17g/mol) = 34g 12. 0.4 moles of NaCl to grams (0.4moles)(58g/mol) = 23.2g 13. 0.8 moles of CO to particles 0.8moles x 6.02x1023 parts/mole 4.82x1023parts 17. 234.0 grams of NaCl to particles 14. 59.5 grams of NH3 to moles 59.5g = 3.5moles 17g/mol 15. 3 moles of CO to liters (3moles)(22.4L/mol) = 67.2L 16. 4.5 moles of C2H4 to liters (4.5moles)(22.4L/mol) = 100.8L 19. 11.2 liters of CO to moles 234.0g = 4.03mol 58g/mol 11.2L = 0.5mol 22.5L/mol 4.03mol x 6.02x1023parts/mol 2.43 x 1024parts 18. 3.20x1025 particles of CH4 to grams 3.20x1025 particles = 53.16mol 6.02 x 1023parts/mol 20. 89.6 liters of CH4 to moles 89.6 liters = 4moles 22.4L/mol (53.16mol)(16g/mol) = 850.56g 21. number of atoms of oxygen in 18.0 grams of water (0.8889)(6.02x1023parts/mol) = 5.35x1023 parts oxygen 16 = 88.89% oxygen in 1 mole H2O 18 22. number of atoms of hydrogen in 18.0g of water 2 = 11.11% hydrogen in 1 mole H2O 18 (0.1111)(6.02x1023) = 6.69x1022parts Empirical Formulas: elements in a chemical formula are in the lowest possible ratios Determine the empirical formulas for each of the following molecular formulas: 33. C8H18 C4H9 38. H2O 34. H2O2 HO 39. C4H8 35. Hg2Cl2 HgCl H2O CH2 40. C7H12 C7H12 CH2 36. C3H6O3 CH2O 41. C4H8 37. Na2C2O4 NaCO2 42. CH3COOH CH2O Steps to find the molecular formula from the empirical formula and the molecular mass: Step 1: determine the mass of the empirical formula Step 2: divide the molecular mass by the empirical formula mass to find the multiple Step 3: multiple the subscripts in the empirical formula by the multiple to find the molecular formula Determine the molecular formula for each of the following: 43. Find the molecular formula for a compound with a mass of 78 amu and an empirical formula of CH. 1(12) + 1(1) = 13g 78 = 6 13 CH C6H6 44. Find the molecular formula for a compound with a mass of 82 amu and an empirical formula of C3H5. 3(12) + 5(1) = 41g 82 = 2 41 C3H5 C6H10 45. Find the molecular formula for a compound with a mass of 90 amu and an empirical formula of HCO2. 1(1) + 1(12) + 2(16) = 45g 90 = 2 45 HCO2 H2C2O4 46. Find the molecular formula for a compound with a mass of 112 amu and an empirical formula of CH2. 1(12) + 2(1) = 14g 112 = 8 14 CH2 C8H16 47. Find the molecular formula for a compound with a mass of 40 amu and an empirical formula of C3H4. 3(12) + 4(1) = 40g 40 = 1 40 C 3 H 4 C3H 4 48. What is the definition of percent? part whole = % Calculate the percent composition of the following compounds: 49. HC2H3O2 1(1) + 2(12) + 3(1) + 2(16) = 60g % H: 4/60 = 6.67% % C: 24/60 x 100 = 40.00% % O: 32/60 = 53.33% 50. NaOH 1(23) + 1(16) + 1(1) = 40g % Na: 23/40 = 57.50% % O: 16/40 = 40.00% % H: 1/40 = 2.50% 51. Calculate the percent of water in compound CaCl210H2O 1(40) + 2(35) + 10(18) = 290g H 2O 180 x 100 = 62.07% H2O 290 52. Circle the compound which contains the largest percentage of nitrogen? Ca(NO3)2 1(40) + 2(14) + 6(16) = 164g 28 x 100 = 17.07% 164 (NH4)2SO4 2(14) + 8(1) + 1(32) + 4(16) = 132g 28 x 100 = 21.21% 132 Regents Type Multiple Choice Review Questions: Q# 1 2 3 4 5 6 answer C C C B B D Q# 7 8 9 10 11 12 answer A A C A C C Q# 13 14 15 16 17 33 answer 1.24moles B A B C D Q# 34 35 36 37 38 39 answer D B D A B B









