Finding Empirical Formulas
advertisement

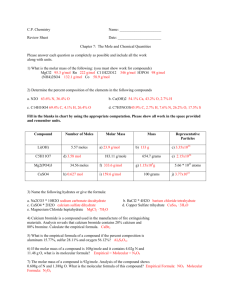
Finding Empirical Formulas Empirical Formula – simplest whole number ratio of moles of the elements in a compound How to find the Empirical Formula: Percent to Mass (change the percentages directly to masses, ie 46% = 46 g) Mass to Moles (Convert the mass to moles – divide by molar mass for that element) Divide by smallest (divide all the mole values by the smallest mole value in the formula) – You can round to whole number AFTER this step. If the decimal is LESS THAN .15 round down. If the decimal is GREATER THAN .85 round up. Multiply until whole (If you cannot round using the rules above find a multiplier that will give you a whole number. Multiply ALL values by the SAME factor until any decimals can easily be rounded to a whole number – round decimals greater than .8 at this time) Molecular Formulas Two or more substances with distinctly different properties can have the same percent composition and the same empirical formula – this simplest whole number ratio of moles of elements does not always indicate the actual number of moles in the compound. The molecular formula distinguishes the substances further. Molecular Formula – specifies the actual number of atoms of each element in one molecule or formula unit of the substance. How do we determine the molecular formula? The molar mass must be determined through experimentation and compared with the mass represented by the empirical formula. Experimentally determined molar mass of the compound = n Mass of the empirical formula of the compound A molecular formula can be determined by multiplying the empirical formula by an integer n Molecular formula = (empirical formula) n The integer is the factor in which the subscripts in the empirical formula must be multiplied to obtain the molecular formula Example – Analysis of a chemical used in photographic developing fluid indicates a chemical composition of 65.45% C, 5.45% H, and 29.09% O. the molar mass is found to be 110.0 g/mol. Determine the molecular formula. 65.45 g C 5.45 g H 29.09 g O 1 mol = 5.44 mol C = 2.91 mol C = 3 12.01 1 mol = 5.40 mol H = 2.89 mol H = 3 1.01 g 1 mol = 1.87 mol O = 1 16.00 g C3H3O = 3(12.01) + 3(1.01) + 16 = 55.06 110.0 ÷ 55.06 = 1.997 = 2 Molecular Formula -- 2(C3H3O) = C6H6O2