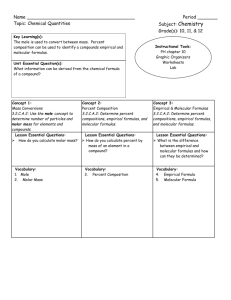
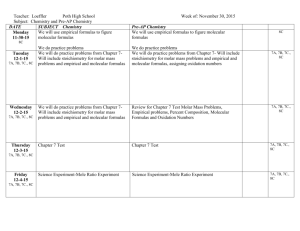
Chapter 4 In-Class Exercises

A 24.7-g sample of limestone, CaCO
3
, contains 9.88 g of calcium. What is the percent Ca in limestone?
A 3.75-g sample of limestone, CaCO
3
, contains 1.80 g of oxygen and 0.450 g of carbon. What is the percent O and the percent C in limestone?
4.17
How many NH
3
molecules are in 0.50 mol of NH
3
?
How many nitrogen atoms and hydrogen atoms are there?
4.18
How many SO
2
molecules are in 1.75 mol of SO
2
? How many sulfur atoms and oxygen atoms are there?
4.24
Calculate the molar mass of each of the following compounds.
(a)
(b)
Hg2Cl2
CaSO4
(c) Cl2O5
⋅
2H2O (d) NaHSO4
4.36
4.37
Calculate the number of moles in 10.0 g of the following substances.
(a)
KHCO
3
(c) Se
(b)
H
2
S
(a)
(d) MgSO
4
Calculate the number of moles in 100.0 g of each of the following compounds.
SO
2
(c) BaSO
4
(b)
Na
2
SO4 (d) KAl(SO
4
)
2
4.57
Given the following molecular formulas, write the empirical formulas.
(a)
C6H4Cl2
(b) C6H5Cl
(c) N2O5
Given the following molecular formulas, write the empirical formulas.
(b)
N2O4 (b) H2C2O4 (c) CH3CO2H
4.58
What are the empirical formulas of the compounds with the following compositions?
(a) 72.36% Fe, 27.64% O
(b) 58.53% C, 4.09% H, 11.38% N, 25.99% O
4.59
What are the empirical formulas of the compounds with the following compositions?
(a) 85.62% C, 14.38% H
(b) 63.15% C, 5.30% H, 31.55% O
A compound with the empirical formula CH2O has a molar mass of approximately 90 g/mol. What is its molecular formula?
A gaseous compound has the empirical formula NO
2
. If its molar mass is approximately 92 g/mol, what is its molecular formula?
A compound has a molar mass of approximately 180 g/mol and a percent composition of 40.00% C, 6.72% H, and 53.29% O.
What is the molecular formula?
A compound with the molar mass of approximately 142 g/mol has the composition 50.7% C, 9.9% H, and 39.4% N. What is its molecular formula?
4.101
Calculate the molarity of each of the following solutions.
(a)
6.30 g HNO
3
dissolved in 255 mL of solution
(b)
49.0 g H
2
SO
4
dissolved in 125 mL of solution
(c)
2.80 g KOH dissolved in 525 mL of solution
(d)
7.40 g Ca(OH)
2
dissolved in 200.0 mL of solution
4.102
How many moles of Na
2
SO
4 are present in 150.0 mL of a 0.124 M solution? How many moles of
Na + ions are
How much water must be added to 935.0 mL of 0.1074 M HCl to obtain a solution that is exactly 0.1000 M?
4.110
If you wish to prepare a 0.055 M solution of NaNO
3
, to what volume would you have to dilute 25 mL of 3.0 M NaNO
3
?
4.111
Determine the concentrations of the following diluted solutions.
(a)
24.75 mL of 0.1832 M HCl diluted to 250.0 mL
(b)
125 mL of 1.187 M NaOH diluted to 0.500 L
(c)
10.00 mL of 0.2010 M acetic acid, CH
3
CO
2
H, diluted to 50.00 mL