Ionic Bonds
advertisement
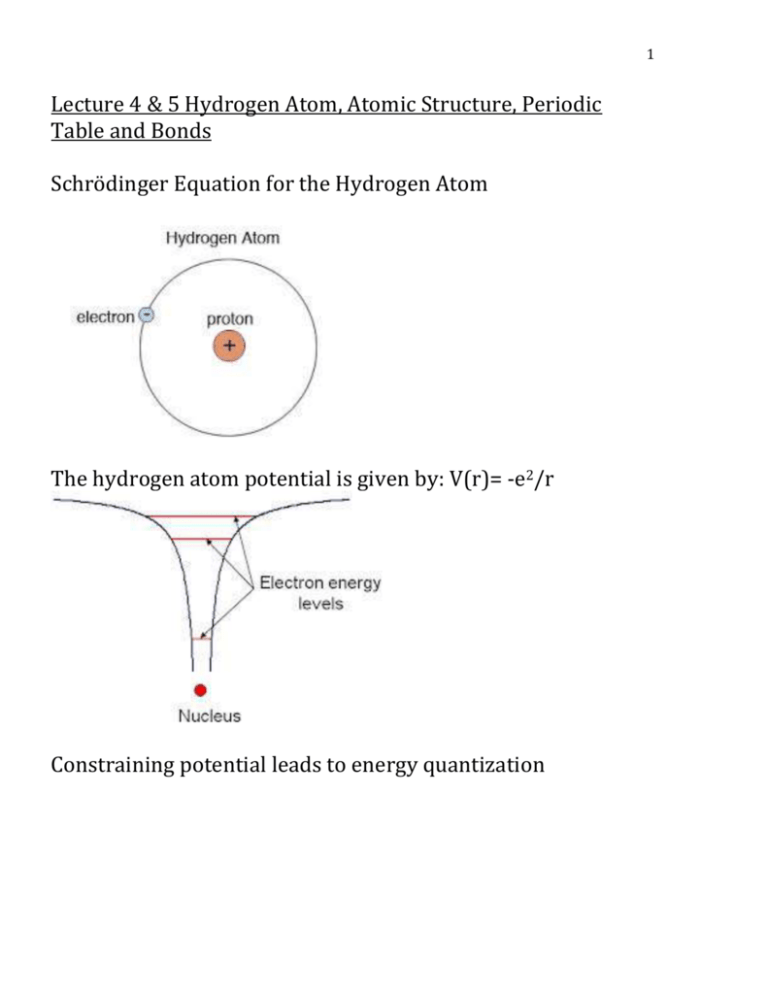
1 Lecture 4 & 5 Hydrogen Atom, Atomic Structure, Periodic Table and Bonds Schrödinger Equation for the Hydrogen Atom The hydrogen atom potential is given by: V(r)= -e2/r Constraining potential leads to energy quantization 2 Applying Sch. eq≅ to find: What is the probability that the electron is found at a distance r from the proton? What are the allowed energy levels The Wave function is a function of time: Ψ(r, t) = ψ(r)𝑤 (𝑡) But if we are interested in how a hydrogen atom behaves on the average |𝑤(𝑡)|2 = 1 We need only to solve time independent sch. Eq. −ℎ2 ( 2𝑚 ∇2 + 𝑉) 𝜓 = 𝐸𝜓 −𝑒 2 Coulomb Potential 𝑉 = 4𝜋𝜖 0𝑟 ℎ2 2𝑚 𝑒2 2 ∇ 𝜓 + (4𝜋𝐸 𝑟 + 𝐸) 𝜓 = 0 1 0 The term r does not lead to analytical solution The radial dependence of the potential suggests that we should transform from Cartesian coordinates to spherical coordinates: (x, y, z)⟶ (𝑟, θ, φ) 3 0£r£¥ p -p £q £ 2 2 0 £ f £ 2p Rotation ×in × xy Rewriting sch eq ≅ in terms of spherical coordinates, (𝑟, 𝜃, 𝜑) it gives 3 diff. eq≅ one in r, and the other two in 𝜃, 𝜑 The solution is a product of the three solutions: Ψ(𝑟, 𝜃, 𝜑 ) = 𝑅 (𝑟)𝜃(𝜃)𝜑(𝜑) Let’s look to spherically symmetrical case, when 𝜓 does not depend on 𝜃, 𝜑 So, we are interested in the radical part only: 4 2 ∇ 𝜓= 𝛿𝜓 = 𝛿𝑥 𝛿2 𝜓 𝛿𝑥 2 𝛿2 𝜓 𝛿𝑦 2 𝛿2 𝜓 + 𝛿𝑦 2 + 𝛿𝑧 2 ∙ 𝛿𝑟 𝛿𝑥 𝛿 𝛿𝜓 𝛿𝑟 = 𝛿𝑥 ( 𝛿𝑟 𝛿𝑥) = ∇ 𝜓= 𝛿𝑧 2 𝛿2 𝜓 𝛿𝑥 2 𝛿𝜓 𝛿𝑟 2 𝛿2 𝑟 𝛿2 𝜓 𝛿2 𝜓 𝛿𝑟 2 𝛿2 𝜓 𝛿𝑧 2 𝛿𝑟 2 = 𝛿𝑟 2 𝛿𝑟 2 [(𝛿𝑥) + (𝛿𝑦) + (𝛿𝑧) ] + 𝛿𝜓 𝛿 2 𝑟 𝛿𝑟 𝛿2 𝑟 (𝛿𝑥 2 + 𝛿𝑦 2 + ) 𝛿2 𝑟 𝛿𝑟 We need to find 𝛿𝑥 and 𝛿𝑥 2 and the other derivatives Straight forward from 𝑟 = √𝑥 2 + 𝑦 2 + 𝑧 2 𝛿2 𝑟 𝛿𝑟 By sub. 𝛿𝑥,… and 2 ∵∇ 𝜓= 𝛿2 𝜓 𝛿𝑟 2 𝛿𝑥 2 in the previous eq. 2 𝛿𝜓 + 𝑟 𝛿𝑟 ⟶ For spherically symmetry Sch. Eq. ℎ2 2𝑚 𝛿2 𝜓 2 𝛿𝜓 𝑒2 ( 𝛿𝑟 2 + 𝑟 𝛿𝑟 ) + (𝐸 + 4𝜋𝜖 𝑟) 𝜓 = 0 0 One solution for this diff eq. ≅ could be 𝜓 = 𝑒 −𝐶0 𝑟 𝐶0 is a constant 5 By differentiating and sub. into equation 1. We can determine the constant 𝐶0 and E. me4 The energy E = − 8ϵ E= 0 2 h2 9.1x1031 (1.6x10−19 )4 − 8(8.85x10−12 )2 (6.63x1034 )2 = −2.178x10−18 J 1𝑒V = 1.6𝑥10−19 𝐽 𝐸 = −13.6𝑒𝑉(It agrees with the lowest energy level of Hydrogen known from spectrum Let’s go back to the other question: What is the probability that the electron can be found between ri r + dr r 2 |ψ|2 = r 2 e−2C0 r since ψ = e−C0 r 6 Corresponding to n= 1 First Bohr orbit More solutions for spherically symmetrical case: ψn = e−C0 r Ln (r) Ln : polynomial The allowed energies: 1 𝐸𝑛 = −13.6 𝑛2 𝑛 = 1,2,3 … n=1 (ground state) n>1 excited states (higher energy, more close to zero) Wave functions for spherically symmetric solutions: -The probability distribution for n>1 have maximum further from the origin. Why? 7 The general solutions (no spherical symmetry) 𝜓𝑛,𝑙,𝑚 (𝑟, 𝜃, 𝜑) The dependence m 3 spatial dimensions lead to 3 quantum numbers n, l, m. n: How far the orbital is? (r) l: How fast the orbit’s angular momentum m: The angle of the orbit in the space They are required to specify the wave function N=1, 2, 3… L=0, 1, 2…(n-1) MI = 0, ±1, ±2 … ± 𝑙 (2𝑙 + 1) 𝐿 𝐿 𝐿 𝐿 =0⟶𝑠 =1⟶𝑝 =2⟶𝑑 =3⟶𝑓 (The usual notation) Sch. Equation for other elements and approximate models Helium atom: 2 protons, 2 neutrons and 2 electrons Disregard neutrons 2 protons, 2 electrons assume the 2 protons are together at the origin For the 2 electrons :∇2 𝜓 ⟶ ∇1 2 𝜓 + ∇2 2 𝜓 P.E. for electron 1: P.E. for electron 2: −2𝑒 2 4𝜋𝐸0 𝑟1 −2𝑒 2 4𝜋𝐸0 𝑟2 8 P.E. of the two electrons: Sch. Eq. 𝑒2 4𝜋𝐸0 𝑟12 r12 : distance between electron 1 and 2 −ℎ2 1 −2𝑒 2 2𝑒 2 𝑒 2 2 2 (∇ 𝜓 + ∇2 𝜓) + ( − + ) 𝜓 = 𝐸𝜓 2𝑚 1 4𝜋𝐸0 𝑟1 𝑟2 𝑟12 No analytical solution! For Sn Z=50 The diff eq≅ have 150 independent variables and 1,275 terms for P.E. Approximate Solutions Assume there are Z protons in the nucleus and Z electrons around the nucleus. The electrons are unaware of each other. The solution for each electron is the same as for Hydrogen atom:____ in H- atom solution, we replace 𝑒 → 𝑍𝑒 2 𝑒 2 → 𝑍 2𝑒 4 The allowed energies are: 𝐸𝑛 = −13.6𝑍 2 𝑛2 (Z, atomic number) We can explain the chemical properties of the elements using this model and we can build up the periodic table from sch. Solution of Hydrogen atoms (with the 3 quantum numbers) 9 Spin quantum number and Pauli exclusion principle Electron spin: electrons demonstrates inherent angular momentum, it is defined by quantum numbers (𝑚𝑠 ) Pauli Exclusion Principle To understand the atomic structure and build the −13.6𝑍 2 periodic table let’s use 𝐸𝑛 = and the 4 quantum 𝑛2 numbers (n, l, 𝑚𝑙 , 𝑚𝑠 ) and the Pauli Principle. * The lowest energy orbital is 1s 10 1 * Energy increase as 𝑛2 *Orbitals of the same n, but different l are considered to be of equal energy, until electron interactions play a role -Build the periodic table: n=1, l=0, 𝑚𝑙 = 0, 𝑠 = ±1 One or two electrons may occupy the lowest energy level, we get hydrogen & He. Note: for He (Helium) shell is closed → This determines why He is chemically inert We go regularly until Potassium then, what? K(Z=19) Because of electron interactions, the energy increases with increasing values of l. For K-element, the energy level of 3d (n=3, l=2) is higher than 4s (n=4, l=0) 11 → 4s shell is filled, then 3d K 4s1 (Notes what happens here) Ca 4s2 Sc 4s23d1 V 4s23d2 Cr 4s13d5 (Note what happens here) Mn 4s23d5 Fe 4s23d6 Co 4s23d7 Cu 4s13d10 Zn 4s23d10 (Note what happens here) Notes that the 4s is the outer shell Rare earth metals: look to their electron configurations and see why it’s hard to distinguish between them chemically. 12 Bonds: What happened when two atoms (2 Hydrogen or 2 Cl) come close to each other? →They form bond to reduce the energy of the system Chemical Bonds (General) 1. 𝐸 → 0 𝑟→∞ 2. 𝑟 → 0 𝐸→∞ 3. Opium Separation: 𝑟0 attractive force= repulsive force 13 Types of Bonding: Ionic Covalent Metallic Van der Waals 1. Ionic Bonds: ex: NaCl Involves the transfer of electrons Na→ loses electron and becomes Na+ Cl→ takes the electron and becomes Cl− - They bond with attraction (covalent) Na+ (positive→cations) Cl− (negative→ anions) (Group I & II): electro positive elements → lose electrons 14 NaCl Bulk Modulus Madelung Energy Q is the charge positive or negative Electrostatic ±𝑞 2 /𝑟 The interaction energy 𝑈𝑖𝑗 𝑈𝑖 = ∑ 𝑈𝑖𝑗 𝑗 𝑟𝑖𝑗 𝑞2 𝑈𝑖𝑗 = 𝜆 exp (− ) ± 𝜌 𝑟𝑖𝑗 𝑅 𝜆 exp (− 𝜌) − 𝑞 2 /𝑟𝑖𝑗 Nearest neighbors 1 𝑞2 ±𝑝 𝑖𝑗 𝑅 otherwise 𝑈𝑡𝑜𝑡 = 𝑁𝑈𝑖 = −𝑅 𝑁(𝑧𝜆𝑒 𝜌 z is the nearest-neighbor contribution 𝛼𝑞 2 − 𝑅 𝜆 and 𝜌 the constants can be determined from gas phase data. 15 𝜌 is the measure of the replusive interaction r= 𝜌 where it is e-1 of the value of r=0. 𝛼= ∑ 𝑗 ± ≡ 𝑀𝑎𝑑𝑒𝑙𝑢𝑛𝑔 𝑐𝑜𝑛𝑠𝑡𝑎𝑛𝑡 𝑝𝑖𝑗 Evaluation of the Madelung Constant: 𝛼 ± = ∑ ≡ 𝑀𝑎𝑑𝑒𝑙𝑢𝑛𝑔 𝑐𝑜𝑛𝑠𝑡𝑎𝑛𝑡 𝑅 𝑟𝑗 𝑗 𝑟 𝑖𝑠 𝑡ℎ𝑒 𝑑𝑖𝑠𝑡𝑎𝑛𝑐𝑒 𝑜𝑓 𝑡ℎ𝑒 𝑗 𝑡ℎ 𝑜𝑛 𝑓𝑟𝑜𝑚 𝑡ℎ𝑒 𝑟𝑒𝑓𝑒𝑟𝑒𝑛𝑐𝑒 𝑖𝑜𝑛 𝑖𝑠 𝑜𝑓 𝑐𝑒𝑛𝑡𝑟𝑎𝑙 𝑖𝑚𝑝𝑜𝑟𝑡𝑎𝑛𝑐𝑒 𝑖𝑛 𝑖𝑜𝑛𝑖𝑐 𝑠𝑜𝑙𝑖𝑑𝑠 𝛼 𝑅 1 = 2 [𝑅 − 1 1 1 + 3𝑅 − 4𝑅 ∙∙∙∙∙∙∙] 2𝑅 or 𝛼 = 2 [1 − 1 1 1 + 3 − 4 ∙∙∙∙∙∙∙] 2 The factor of 2 occurs because there are two ions left and right at equal distance rj 𝑙𝑛(1 + 𝑥 ) = 𝑥 − 𝑥2 2 + 𝑥3 3 − 𝑥4 4 one-dimensional chain is 𝛼 = 2 ln 2 16 Three dimensions series is more difficult. 𝑒2 _ _ ..1 __ • ''"IT l' . ' Electrostatic energy = -M (4𝜋𝜖 𝑎) 0 lattice constant a= 2.8 Å E=8.94 Solutions: U(pot) = N (− Pij = 𝑟𝑖𝑗 𝑟 𝑒2 𝑟 ±1 𝛼 1 Σ 𝑃 + 𝑟𝑛 Σ 𝑃 ) 𝑖𝑗 𝑖𝑗𝑛 r = 2.10 U(r) = N (− 𝑒2 𝑟 𝐶 α + 𝑟𝑚) 𝜕𝑢 𝑚𝐶 𝑚−1 = 0 𝑡ℎ𝑒𝑟𝑒𝑓𝑜𝑟𝑒 𝑟0 = ð𝑟 𝛼𝑒 2 𝛼𝑒 2 𝑟0𝑚−1 𝐶= 𝑚 substituting m ranges 6-9 𝑢0𝑡ℎ = 𝑢(𝑟0 ) = − 𝛼𝑒 2 𝑚−1 𝑟0 ( 𝑚 ) 17 First Sum ( Na+ and Cl-) By definition M = 1.748 Second Sum 1 𝛼Σ 𝑃 = C 𝑖𝑗𝑛 𝛿𝑢 P = -𝛿𝑣 V = 2Nr3 δV = 6Nr2δr 1 𝛿 1 P = -6𝑁𝑟 2 𝛿𝑟 𝑁(−𝑀𝑒 2 𝑟 + 𝑀𝑒 2 1 = -6𝑁𝑟 2 ( 𝑟 𝐶 ) 𝑟𝑛 𝑛𝐶 - 𝑟 𝑛+1 ) P = 0 = same as 1 atm 1 𝑀𝑒 2 𝑟0 𝑟02 2(0) = ( 𝑛𝐶 1 - 𝑟 𝑛+1 ) 𝑟_2 Compressibility B=- 𝑉𝜕𝑃 𝜕𝑉 1 = 2Nr3 𝛿 𝜕 1 𝑀𝑒 2 ( 6𝑁𝑟 2 𝜕𝑟 6𝑟 2 𝑀𝑒 2 = 18 𝑟 𝛿𝑟 ( 1 𝑟4 𝑛𝐶 – 𝑟 𝑛+3 ) 𝑟 𝑛𝐶 - 𝑟 𝑛+1 ) 18 1 Using two Eg.** = 18 ( 𝐶 1 𝑛(𝑛+3) 𝑟0𝑛+3 − 𝑀𝑒 2 4 𝑟04 ) 2 = 𝑀𝑒 𝑛+3 𝑟 𝑛𝑟 4 0 B= 0 𝑀𝑒 2 ( 18 𝑛(𝑛+3) 𝑛 𝑒 2𝐴 Φ coul. = 4𝜋Є 0 𝑟0 1 1 1 = 18 𝑀𝑒 2 𝑟 3 (𝑛 − 1) − 4) 𝑟 4 0 0 = 8.53 𝑒𝑉 18𝑟03 𝐵 n = 1 = Φ coul n= 1+18 (2.82𝑥10−8 𝑐𝑚)3 (2.4𝑥1011 𝑑𝑦𝑛/𝑐𝑚2 ) 8.53𝑥10−12 𝑛𝑐 Φ coul = 𝑟 𝑛 → 𝐶 = 𝑀𝑒 2 𝑟2 0 𝑛𝐶 𝛼𝑒 2 0 𝑟04 = 𝑟𝑛 = = 8.1 Φ coul 𝑟0𝑛 𝑛 C = 1.05eV (44.36Å)8.1 = 4660eV∙A8.1 1 1 1 Σ n = Є + 12 ( 4.05 ) + ( 4.05 ) = 6.81 eij 2 3 𝐶 α = 6.81 = 684eVÅ8.1 −𝛼𝑒 2 ucoul = 𝑟 α=M 𝑛𝐶 𝑟0𝑛−1 = 𝑒 2 𝛼 19 C= 𝑢0𝑡ℎ −𝛼𝑒 2 𝑟0𝑚−1 𝑛 = 𝑢(𝑟0 ) = −𝛼𝑒 2 (𝑛−1) 𝑛𝑟0 n =8.1 18𝐵𝑟03 n = 1 + 1𝑢 𝑐𝑜𝑢𝑙(𝑟0 ) 20 E(n) = energy atom Total E: Na3 E(r0) Na = −𝛼𝑒 2 7.1 𝑟0 (8.0) atoms/cm3 The cohesive energy: 8.13-1.53= 6.60eV with respect Na+,Cl- 21 : The energy required to overcome bonding at T=0K Electro negative elements→ accept electrons Compressed (T) 22 Na a3 E(r0 – ∆r) = Total energy will increase Expanding in a Taylors Series: 𝑑2 𝐸 E (r0) + ½ ( 𝑑𝑟 2 ) ∆𝑟 2 + ⋯ ½ Na a3( 𝑑2 𝐸 ) 𝑑𝑟 2 𝑟=𝑟0 (∆𝑟)2 Eq. 5.2 23 𝑎 ( ) ∆𝑟 → 𝑒𝑎𝑐ℎ 𝑓𝑎𝑐𝑒 𝑐𝑜𝑚𝑝𝑟𝑒𝑠𝑠𝑒𝑑 2𝑟0 T is the stress increasing from 0 to T 6x( 1 𝑇 𝑎2 2 𝑎∆𝑟 Eq. 5.3 ) ( 2𝑟 ) 0 equality Eq. 5.3 and 5.2 Bulk Elastic Modulus ∆𝑎3 T = c 𝑎3 ≅ 1 𝐶 3∆𝑎 𝑎 =𝑐 3∆𝑟 𝑟0 𝜕2𝐸 C = 9𝑟 ( 𝜕𝑟 2 ) 0 𝑟=𝑟0 Hooke’s Law Solutions (Approximate) Z protons in the nucleus Z electrons are the nucleus Free and no interaction 24 𝑒2 𝑟 → 𝑍𝑒 2 𝑟 ucoul → 𝑒2 →𝑟 ! (𝑍 + 𝑒)(𝑍 − 𝑒) 4𝜋Є0 𝑟 = nearly ideal ionic bonds are alkali halides Metals: (**) carriers of the negative charge are highly mobile **electrons work as the glue that holds the lattice together. Strong cohesive energy ductile and malleable Typical bonding per atom Dominant Bonding: Covalent diamond , Metallic Ionic Van der Waals: Cohesive Energy (eV) C.E. S ≈ 4.8 eV/atom Na, Fe, La ≈ 1-9 eV/atom NaCl, MgO ≈ 5 (eV) per ion pair He, Ar, Cl2 ≈ .02 - .03 25 Several aspects of the van der Waals interaction and the resulting bonding should be emphasized: 1. The interaction represented by U(r) is attractive and leads to the formation of a potential well when the repulsive Coulomb interactions between the atoms are also included. 2. The van der Waals interaction is quite weak and is also very short range, due to the 1/r6 dependence of U(r). 3. When an empirical repulsive potential of the form +B/r12 is added to the attractive van der Waals term, the result is known as the Lennardlones potential, Here and are parameters characterizing the strength and range of the interaction, respectively. This potential is often used in calculations of the cohesive energies of inert-gas crystals. ---- 2. Covalent Bonds Involves sharing of electrons, atoms like to have filled shells C 2s22p2 26 Si 3s23p2 Ge 4s24p2 These elements are tetravalent instead of divalent? Why? Look to diamond structure picture: each atom is surrounded by 4 neighbors. Carbon in the form of diamond is: insulator-it has strong covalent bond. Si & Ge: The bonds are weaker, they are semiconductors Carbon in the diamond form is the hardest material (high resistant to deformation) Diamond has a higher thermal conductivity than metals at room temperatures!!!! Why? Metallic Bonds: In metals, the electrons are highly mobile. Sea of electrons acts as a glue that holds the positively charged metal ions from flying apart. Delocalized electrons. 27 Electron charge density of valence band densities between the pair of atoms 3. Van der Waals bonds. (graphite) 28 Instantaneous asymmetric charge distribution in the atoms and molecules→these distributions cause temporary dipole and the atoms or molecules one attracted by electrostatic forces. Derivative A 𝐵 E(r) = n − 𝑚 r 𝑟 𝑚 𝐴= 𝑛 𝑚 𝐵 𝐵 𝐸𝑐 = − 𝑛 𝑟0 𝑚 𝑟0 𝑚 𝐵 𝑚 𝐸𝑐 = 𝑚 ( − 1) 𝑟0 𝑛 𝑚<𝑛 Dipole-dipole bonds 29 When the bond is not exactly ionic or covalent, shared electrons one close to one atom (more electronegative) →bond is polar and the molecule has a dipole moment. Those molecular bonds due to higher electron density on one side of the nucleus 30 Lennord-Jones potentials which describes interaction of two inert Gas atoms such as Ne,Ar, Kr and Xe. Ne Ar Kr Xe R/ Derivative A 𝐵 E(r) = n − 𝑚 r 𝑟 𝑚 𝐴= 𝑛 𝑚 𝐵 𝐵 𝐸𝑐 = − 𝑛 𝑟0 𝑚 𝑟0 𝑚 𝐵 𝑚 𝐸𝑐 = 𝑚 ( − 1) 𝑟0 𝑛 𝑚<𝑛 Message: Nearly ideal ionic bonds are alkali halides. Metals: ** Carriers of the negative charge are highly malleable ** Electrons work as thee glue that holds the lattice together. Strong cohesive energy ductile and malleable. Dominant Bonding: Covalent: diamond, Si, Ge ≈ 4.8 eV/atom 31 Metallic: Na, Fe, La ≈ 1-9eV/atom Ionic: NaCl, MgO≈ 5(eV) per ion pair Van der Waals: He, Ar, Cl2 ≈.02-.03







