Comparisons of Unknowns: Weak Acid Identification
advertisement

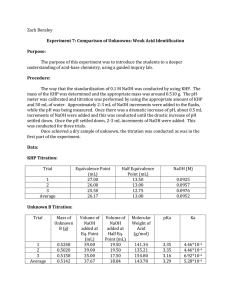
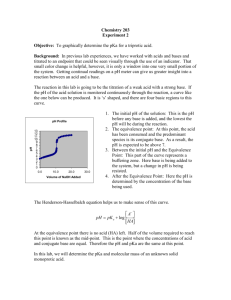
Experiment 7: Comparison of Unknowns: Weak Acid Identification Introduction: This is a guided inquiry lab that will have seven parts. The main goal is to have a better understanding of acid-base chemistry. This is done by following guidelines and answer questions during the lab to get to the final step. Procedure: Standardize 0.1M NaOH using KHP. Weigh out about 0.5106g KHP for a 25mL titration. Calibrate the pH meter and then fill three flasks each with the appropriate amount of KHP and 50mL of water. Then add the NaOH to the flask of KHP in increments of 2-3mL at a time while take pH readings after each addition. Once you notice a dramatic change in pH slow the addition to only 0.5mL and do this until the change slows back down and then you can go back to adding around 2-3mL at once until there is minimal change. Do this technique until you have three good trials. Once you obtain a dry sample of your unknown do the titration in the same manor. This lab was done by following the seven steps handed out in lab and answering the questions during this process. Data: KHP Titration Trial Eq. Point (mL) Half Eq. Point (mL) NaOH (M) 1 25.1mL 12.55mL 0.1000 2 25.1mL 12.55mL 0.1000 3 25.1mL 12.55mL 0.1001 Average 25.1mL 12.55mL 0.1000 Standardizing graphs (3 trials) Unknown C Titration Trial Mass of Unk C (g) Volume NaOH Volume NaOH added at Eq. added at Half Point (mL) Eq Point (mL) MW Acid (g/mol) pKa Ka 1 0.3072g 18.2 9.1 168.791 3.9 1.26 ∗ 10−4 2 0.3078g 18.1 9.05 170.055 4.1 7.9 ∗ 10−5 3 0.3076. g 18.0 9 170.889 4.0 1.00 ∗ 10−4 18.1 9.05 169.912 4.0 1.02 ∗ 10−4 Average Unknown Acid C titrations (3 trials) Calculations: Mass of KHP needed 1𝑚𝑜𝑙𝐾𝐻𝑃 204.23𝑔 𝑉𝑁𝑎𝑂𝐻 ∗ 𝑀 ∗ ∗ = 𝑔𝐾𝐻𝑃 1𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 1𝑚𝑜𝑙 𝐾𝐻𝑃 Molarity of NaOH 1 mol KHP 1 mol NaOH 1 mol NaOH g KHP x x x = M NaOH 204.23g 1 mol KHP 0.0251 L Moles of Unknown Acid 1 𝑚𝑜𝑙 𝑢𝑛𝑘 𝑎𝑐𝑖𝑑 𝑉 𝑁𝑎𝑂𝐻 ∗ 𝑀 ∗ = 𝑚𝑜𝑙 𝑢𝑛𝑘 𝑎𝑐𝑖𝑑 1 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 MW of Unknown Acid 𝑔 𝑈𝑛𝑘𝑜𝑤𝑛 𝑎𝑐𝑖𝑑 = 𝑀𝑊 𝑚𝑜𝑙 𝑢𝑛𝑘𝑜𝑤𝑛 𝑎𝑐𝑖𝑑 pKa of Unknown Acid C 1 𝑝𝑘𝑎 = 𝑝𝐻 𝑎𝑡 𝑉𝑒 2 Ka of Unknown Acid C 𝐾𝑎 = 10−𝑝𝑘𝑎 Mass of KHP needed Example 0.025𝐿 ∗ 0.1𝑀 ∗ 1𝑚𝑜𝑙𝐾𝐻𝑃 204.23𝑔 ∗ = 0.5106𝑔𝐾𝐻𝑃 1𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 1𝑚𝑜𝑙 𝐾𝐻𝑃 Molarity of NaOH trial 1 0.5126g KHP x 1 mol 1 mol 1 mol x x = 0.1000M NaOH 204.23g 1 mol 0.0251 L Moles of Unknown Acid trial 1 1 𝑚𝑜𝑙 𝑢𝑛𝑘 𝑎𝑐𝑖𝑑 1 𝑚𝑜𝑙 𝑁𝑎𝑂𝐻 = .00182𝑚𝑜𝑙 MW of Unknown Acid Trial 1 0.3072𝑔 𝑈𝑛𝑘𝑜𝑤𝑛 𝑎𝑐𝑖𝑑 = 168. 791𝑀𝑊 0.00182𝑚𝑜𝑙 𝑢𝑛𝑘𝑜𝑤𝑛 𝑎𝑐𝑖𝑑 pKa of Unknown Acid C Trial 1 3.9 = 3.9 . 0182𝐿 𝑁𝑎𝑂𝐻 ∗ 0.100𝑀 ∗ Ka of Unknown Acid C Trial 1 1.26 ∗ 10−4 = 10−3.9 Possible Identities of Unknown Acid C: -1-Naphthoic Acid: 172.18g/mol and Ka=2.1x10-4 -Acetylsalicylic Acid: 180.157g/mol and Ka=3.3x10-4 When looking at our data the formula weight is very close to 1-Naphthoic Acid but the pka we found, 4, is a little higher than the expected 3.68. This could be due to estimating with the graph and not getting an exact equivalence point and not getting an exact half equivalence point. I probably should have point more points on the graph and printed it out in order to make it more accurate. For Acetylsalicylic acid our formula weight and pKa and Ka are even further off from the expected values. Also another error could have been with the pH meter and not waiting for it to stabilize so my pHs could be off during my actual titration. We found that weak acids complicate calculating pH at equivalence point because you need to use and ice chart which is not needed when working with strong acids and bases. By looking at the graph it can help you determine both your Equivalence point and your half equivalence point which can help determine the pKa. The volume of titrant added halfway up the jump is roughly your equivalence point. When that volume is divided in half you have your half equivalence point. When looking at the graph whichever pH corresponds to the volume at the half equivalence point is the pKa. This is helpful when trying to determine the identity of the unknown acid because you have no molar mass or Ka to work with.