Cells and Batteries Lab - University of Michigan SharePoint Portal
advertisement

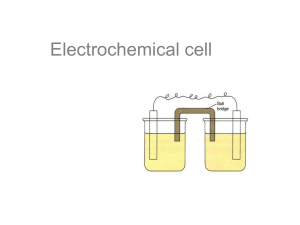
Electrochemical Cells and Batteries UM Physics Demo Lab 07/2013 In the static electricity lab we saw that separated charges travel to recombine to a state of charge neutrality. The charges were separated by rubbing two materials together (triboelecticity) or by polarizing a conductor. Another way to separate charges is electrochemically, and this is the principal that allows us to chemically generate electrical energy in a non-rechargeable electrochemical cell and to store and retrieve electrical energy in a rechargeable electrochemical cell. An electrochemical cell is composed of three components: a cathode, an anode, and an electrolyte solution. See figure below. Figure 1: Schematic of a commercial cell A collection of cells connected together is called a battery. Usually the cells are connected in series to increase the available electrical potential (voltage) compared to a single cell. Sometimes cells are connected in parallel to increase the capacity (ability to deliver current) of the battery when compared to a single cell. Battery Acid An essential feature of a battery is the electrolyte solution that connects two electrodes. Coke, lemons, and potatoes are fun materials that can be used, but powerful acids or bases are more commonly used for commercial purposes because they produces cells with a higher capacity to deliver current. When the anode and cathode are immersed in the electrolyte, a chemical reaction takes place. The result of that reaction is a shortage of electrons on the cathode and a buildup of electrons on the anode. This charge imbalance makes the electrons want to travel from the anode to the cathode. The elegance of an electrochemical cell is that the chemistry of the electrolytic solution has a preferential current direction for a given voltage, and won’t allow the electrons to flow back to the cathode within the cell. For the charge imbalance to neutralize, the current must travel through an external circuit. The traveling electrons are what power your electronic device. Property of LS&A Physics Department Demonstration Lab Copyright 2006, The Regents of the University of Michigan, Ann Arbor, Michigan 48109 1 Traveling charges are called a current. The current is determined by two things. 1. The cell has a maximum electric potential or voltage (defined as the potential energy available per unit of charge) that depends on how it is built and how much of the chemical reactants have been consumed. Electric potential is also sometimes referred to as “Electromotive Force” or EMF. 2. The resistance in the external circuit also determines the current. Electrical current is much like a current of water. A pump gives elevation to the water as a cell gives voltage to electrons at the anode. The flow of water to lower elevation is analogous to the current flowing in the circuit from higher electrical potential energy to lower electrical potential energy and the constrictions in the water’s path are analogous to the circuit’s resistance. Property of LS&A Physics Department Demonstration Lab Copyright 2006, The Regents of the University of Michigan, Ann Arbor, Michigan 48109 2