polb23406-sup-0001-suppinfo
advertisement

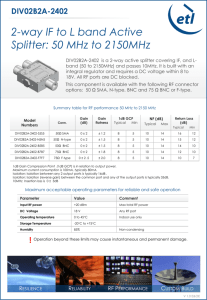
SUPPLEMENTARY INFO RYodx38 Current (A) 0.0002 0.0000 -0.0002 -0.0004 -2.0 -1.5 -1.0 -0.5 0.0 0.5 1.0 1.5 Potential (V) Figure1. Working electrode: Glassy Carbon (drop coating) Counter: Pt wire Reference: Ag/AgCl Electrolyte : 0.1M TBAPF6 in acetonitrile (tetrabutylammonium hexafluorophosphate) Copolymer EHOMO: -5.40 eV Copolymer ELUMO: -2.68 eV B 0.0003 0.0002 B 0.0001 0.0000 -0.0001 -0.0002 -0.4 -0.2 0.0 0.2 0.4 0.6 0.8 A Figure2. Ferrocene/ 0.1M TBAPF6/AN 50mV/s Eonset: 0.386 Instrumentation: Cyclic voltammetry (CV) measurements were performed on a potentiostat/galvanostat model EPP4000 (Princeton Applied Research) using an Ag/AgCl reference electrode, a platinum wire counter electrode, and a platinum foil working electrode. The working electrode was coated with a polymer film by drop-casting a polymer solution in chloroform. CV measurements were recorded in 0.1 M tetrabutylammonium hexafluorophosphate in dry acetonitrile at a sweeping rate of 50 mV.s -1using ferrocene/ferrocenium (Fc/Fc+) couple as a standard. The HOMO energy levels were calculated using the equation: EHOMO (eV) = - (Eoxonset- EFc/Fc+onset) - 4.80 eV where Eoxonset and EFc/Fc+onset are the onset oxidation potentials for the polymer sample and the ferrocene against the Ag/AgCl reference electrode, while the value – 4.8 eV is the HOMO energy level of ferrocene against vacuum1. The LUMO energy levels were calculated with the HOMO levels obtained by CV measurements and the optical band gap (Egopt) obtained by UV-Vis measurements: ELUMO (eV) = Egopt+ EHOMO. 0,08 a D0 D1 D2 D3 D4 D5 D6 External (QE [%]) 0,06 0,04 0,02 0,00 4 6 8 10 12 14 16 Voltage [V] D0 with ETL D1 with ETL D2 with ETL D3 with ETL D4 with ETL D5 with ETL D6 with ETL 2 External (QE [%]) b 1 0 4 6 8 10 12 14 16 Voltage (V) Figure 3. External Quantum Efficiency – Voltage graphics of the devices a) with and b) without ETL containing QDs with different concentrations. Reference 1. (a) M. Kumada and K. Tamao, Adv. Organomet. Chem.1968, 6, 19. (b) J. Pommerehne, H. Vestweber, W. Guss, R. F. Mahrt, H. Bassler, M. Porsch and J. Daub, Adv. Mater. 1995, 7, 551. (c) B. W. D. Andrade, S. Datta, S. R. Forrest, P. Djurovich, E. Polikarpov and M. E. Thompson, Org. Electron. 2005, 6, 11.