2013
advertisement

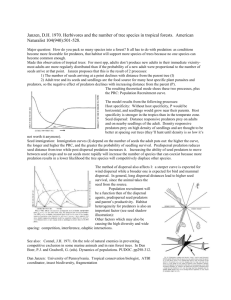
Chris Jeffs DPhil Student – University of Oxford VG Fellowship Insect pre-dispersal seed predation across a tropical rainfall and plant species richness gradient Tropical forests support an exceptional richness of flowering plants, many with over 250 tree species per hectare1. But how are so many ecologically similar species competing for the same limiting resources (light, water, nutrients) able to coexist? The Janzen-Connell mechanism proposes that negative density-dependent mortality mediated by natural enemies, such as pathogens and herbivores, prevents a small number of dominant competitors from monopolising space. This prevents the exclusion of weaker and rarer species, thus enhancing diversity2,3,4. Recent survey work in the lowland tropical forest on Barro Colorado Island, Panama, has shown that insect seed predators consuming seeds pre-dispersal show high levels of host-specificity5. Specialisation is an essential factor in the Janzen-Connell mechanism as it ensures the creation of enemy-free space for other, rarer species to occupy. However, pre-dispersal seed predators are rarely studied, and assessments of their specificity across larger spatial scales and climatic gradients are unknown. With evidence that pathogen and herbivore activity increases positively with humidity6,7, it is possible that globally observed positive correlations between rainfall and species richness8,9 may be driven by more intense natural enemy pressure as rainfall increases. The 80 km stretch of rainforest spanning the Isthmus of Panama demonstrates such a steep increase in both humidity and species richness when moving from the drier Pacific to the more humid Atlantic coast10. Between March and June 2014 I travelled to Panama to investigate how seed predators may contribute to the structuring of diverse tropical forest communities. Conducting experiments that contributed to work at Oxford University led by Dr Owen Lewis, Dr Lars Markesteijn and Patrick Kennedy, we assessed whether: 1) Mean pre-dispersal seed predation rates increase with humidity 2) Prevalence and strength of enemy mediated negative density-dependent mortality increases with humidity 3) Host-specificity of pre-dispersal insect seed predators increases with humidity Freshly abscised seeds were collected from target tree species and families at eight 1 ha mapped forest plots spanning the rainfall and species richness gradient in order to rear seed predators developing within them. Emerging adults and larvae were pinned or stored in alcohol for later morphological identification and/or DNA barcoding to allow the construction of a plant-predator food web to assess specificity variation across the gradient. After a three month rearing period, seeds were dissected to estimate mean predation rates for focal plant species, families and sites spanning the rainfall gradient. By the completion of the 2014 field season, 33,393 seed Chelofonyx sp., (Coleoptera:Curculionidae) seed dispersal units were collected from 51 plant species, with a total predator of Quararibea asterolepis (Malvaceae). of 47 seed predator morphospecies reared. It now remains to Copyright and photo credit Indira Simon. have all insect material identified, but preliminary assessments appear to corroborate evidence of high host-specificity in predispersal insect seed predator food webs, with a single host plant for 91.5% of morphospecies. Cross-gradient analyses of specificity and predation rates remain to be conducted, but hopefully will yield exciting results. I would like to thank the board of the Varley-Gradwell fellowship for supporting my research, and would strongly encourage others to apply. I am very grateful for the experience this funding has given me. References 1. Valencia R, et al. in Tropical Forest Diversity and Dynamism (eds. Losos, EC, et al.) (University of Chicago Press, 2004). 2. Harms, K.E., et al. Nature 404, 493-495 (2000). 3. Petermann, J.S., et al. Ecology 89, 2399-2406 (2008). 4. Comita, L.S., et al. Science 329, 330-332 (2010). 5. Gripenberg, S., unpublished data. 6. Coley, P.D. Clim. Change 39, 455-472 (1998). 7. Brenes-Arguedas, T., et al. Ecology 90, 1751-1761 (2009). 8. Phillips, O.L., et al. Proc. Natl. Acad. Sci. U. S. A. 91, 2805-2809 (1994). 9. Kreft, H., et al. Proceedings of the National Academy of Sciences 104, 5925-5930 (2007). 10. Engelbrecht, B.M.J., et al. Nature 447, 80-82 (2007).