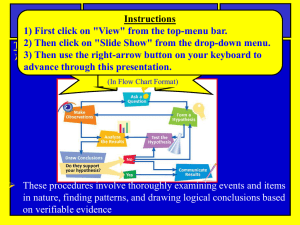
Curriculum and Instruction * Office of Science
advertisement

Introduction In 2014, the Shelby County Schools Board of Education adopted a set of ambitious, yet attainable goals for school and student performance. The District is committed to these goals, as further described in our strategic plan, Destination2025. By 2025, 80% of our students will graduate from high school college or career ready 90% of students will graduate on time 100% of our students who graduate college or career ready will enroll in a post-secondary opportunity In order to achieve these ambitious goals, we must collectively work to provide our students with high-quality, College and Career Ready standards-aligned instruction. Acknowledging the need to develop competence in literacy and language as the foundation for all learning, Shelby County Schools developed the Comprehensive Literacy Improvement Plan (CLIP). The CLIP ensures a quality balanced literacy approach to instruction that results in high levels of literacy learning for all students across content areas. Destination 2025 and the CLIP establish common goals and expectations for student learning across schools. CLIP connections are evident throughout the science curriculum maps. The Tennessee State Standards provide a common set of expectations for what students will know and be able to do at the end of a grade. College and Career Ready Standards are rooted in the knowledge and skills students need to succeed in postsecondary study or careers. While the academic standards establish desired learning outcomes, the curriculum provides instructional planning designed to help students reach these outcomes. Educators will use this guide and the standards as a roadmap for curriculum and instruction. The sequence of learning is strategically positioned so that necessary foundational skills are spiraled in order to facilitate student mastery of the standards. Our collective goal is to ensure our students graduate ready for college and career. The standards for science practice describe varieties of expertise that science educators at all levels should seek to develop in their students. These practices rest on important “processes and proficiencies” with longstanding importance in science education. The Science Framework emphasizes process standards of which include planning investigations, using models, asking questions and communicating information. 2015-2016 1 Curriculum and Instruction – Office of Science Middle School Science: Map for Grade 8 First Nine Weeks Construct explanations and design solution Obtain, evaluate, and communicate information Engage in argument Ask questions and define problems Patterns Develop and use models Practices in Science Use math, technology, and computational thinking Plan and carry out investigations Cause and Effect Stability and change Cross Cutting Concepts Analyze and interpret data Energy and matter Systems and system models Crosscutting concepts have value because they provide students with connections and intellectual tools that are related across the differing areas of disciplinary content and can enrich their application of practices and their understanding of core ideas. Throughout the year, students should continue to develop proficiency with the eight science practices. Crosscutting concepts can help students better understand core ideas in science and engineering. When students encounter new phenomena, whether in a science lab, field trip, or on their own, they need mental tools to help engage in and come to understand the phenomena from a scientific point of view. Familiarity with crosscutting concepts can provide that perspective. A next step might be to simplify the phenomenon by thinking of it as a system and modeling its components and how they interact. In some cases it would be useful to study how energy and matter flow through the system, or to study how structure affects function (or malfunction). These preliminary studies may suggest explanations 2015-2016 2 Curriculum and Instruction – Office of Science Middle School Science: Map for Grade 8 First Nine Weeks for the phenomena, which could be checked by predicting patterns that might emerge if the explanation is correct, and matching those predictions with those observed in the real world. Science Curriculum Maps This curriculum map is designed to help teachers make effective decisions about what science content to teach so that, our students will reach Destination 2025. To reach our collective student achievement goals, we know that teachers must change their instructional practice in alignment with the three College and Career Ready shifts in instruction for science. To ensure that all student will be taught science content and processes in a comprehensive, consistent, and coherent manner, Science Curriculum Maps are provided. Foundation texts for the maps include Shelby County Schools Framework for Standards Based Curriculum, Science Curriculum Frameworks-K-12 (State of Tennessee Board of Education, and National Science Education Standards). Teachers function most effectively and students learn best within an “aligned” curriculum delivery system. An aligned system begins with a concerted effort to implement the state curriculum frameworks. Many districts have developed curriculum guides built around these frameworks to ensure that what is taught in particular grades and courses is closely linked with student Learning Expectations found in the state standards. Classroom teachers use these locally-generated curriculum guides to plan and implement their individual grade or course Pacing Guides. Expectations for student performance are clear and carefully tied to daily instructional events and classroom assessment practices. In theory, a fully aligned system closes the loop between state standards and student learning. Additionally, a coherent instructional/assessment system offers the potential for heightening student learning as reflected by their performance on state-mandated standardized tests. Our collective goal is to ensure our students graduate ready for college and career. Most of the elements found in the state Curriculum Frameworks were incorporated into the curriculum mapping materials prepared by Shelby County Schools. Additional features were included to add clarity and to offer avenues that could assist teacher in developing grade level lessons. A district-wide, K-12, standards-based curriculum is implemented in science. This curriculum is articulated in the form of individual SCS curriculum maps for each grade and subject. These SCS curriculum maps enable the district to implement a single curriculum that emphasizes specific standards. Since Shelby County has a high rate of mobility among the student population, the SCS curriculum maps ensure that all students receive the same program of high-level instructional content and academic expectations, 2015-2016 3 Curriculum and Instruction – Office of Science Middle School Science: Map for Grade 8 First Nine Weeks regardless of which school they attend. The utilization of a district-wide standards-based curricular program ensures that students in SCS are engaged in hands-on inquiry based activities as teachers implement the cu 2015-2016 4 Curriculum and Instruction – Office of Science Middle School Science: Map for Grade 8 First Nine Weeks State Standards Embedded Standards Learning Outcomes Resources* Core Ideas Unit 1.1 Properties of Matter - 3 weeks Academic vocabulary: atom, atomic mass, atomic number, chemical change, density GLE 0807.9.2: Explain that matter has properties that are determined by the structure and arrangement of its atoms. GLE 0807.Inq.2: Use appropriate tools and techniques to gather, organize, analyze, and interpret data. Use appropriate tools and techniques to measure mass, volume, and temperature of matter. GLE 0807.9.3: Interpret data from an investigation to differentiate between physical and chemical changes. GLE 0807.Inq.1: Design and conduct open-ended scientific investigations. Design and conduct investigations to determine the physical and chemical properties of matter. GLE 0807.Inq.5: Communicate scientific understanding using descriptions, explanations, and models. GLE 0807.Inq.3 Synthesize information to determine cause and effect relationships between evidence and explanations. Calculate the density of matter using data collected in an investigation. Compare densities of matter, using a density column, tank of water, and mathematics. Tennessee Holt Science and Technology TE, Chapter 7 Section 1: What is Matter? and Section 2: Physical Properties and Section 3: Chemical Properties p. 180-199. Glencoe Tennessee Science 8; Chapter 4 p. 98-125; Chapter 5 126-151. STCMS Properties of Matter TG: Lessons 2-4, p. 15-48. STCMS Properties of Matter Content related real-life articles SG • Archimedes' Crowning Distinguish between physical and Moment pp. 22-23 • Why did the Titanic chemical changes. Float? p. 28-29 • Deadly Density p. 34 • Air Heads p.35-37 NGSS Practice 3: Planning and carrying out investigations NGSS Practice 4: Analyzing and interpreting data NGSS Practice 5: Using mathematics and computational thinking Writing: Support claims with logical reasoning and relevant, accurate data and evidence that demonstrate an understanding of the topic or text, using credible sources. Reading: Follow precisely a multistep procedure when carrying out experiments, taking measurements, or performing technical tasks. 2015-2016 5 Curriculum and Instruction – Office of Science Middle School Science: Map for Grade 8 First Nine Weeks Writing: Use precise language and domain- specific vocabulary to inform about or explain the topic Unit 1.2 Phases of Matter, 2 weeks Academic vocabulary: solid, liquid, gas, mixture, pressure, temperature, volume GLE 0807.9.1: Understand that all matter is made up of atoms. GLE 0807.9.2: Explain that matter has properties that are determined by the structure and arrangement of its atoms. GLE 0807.9.3 Interpret data from an investigation to differentiate between physical and chemical changes. GLE 0807.Inq.2: Use appropriate tools and techniques to gather, organize, analyze, and interpret data. GLE 0807.Inq.1: Design and conduct open-ended scientific investigations. GLE 0807.Inq.5: Communicate scientific understanding using descriptions, explanations, and models. GLE 0807.Inq.3 Synthesize information to determine cause and effect relationships between evidence and explanations. GLE 0807.9.5: Apply the chemical properties of the atmosphere to illustrate a mixture of gases. GLE 0807.Inq.4 Recognize Design and conduct investigations to determine the effects of changes in temperature, pressure and volume on the density and state of matter. Tennessee Holt Science and Technology TE, Chapter 8, Section 1: Three States of Matter and Section 2: Behavior of Gases and Section 3: Changes of State p. 208 - 223 Evaluate the relationships between pressure, volume and temperature of a gas. Glencoe Tennessee Science 8; Chapter 6 p. 152-183. Construct graphics that illustrate the energy changes during a phase change. Diagram and explain the behavior of particle movement in solids, liquids, and gases. Explain how the chemical STCMS Properties of Matter TG: Lessons 6 – 9, p. 65 - 109. NGSS Practice 3: Planning and carrying out investigations NGSS Practice 2: Developing and using models NGSS MS-PS1-4: Develop a model that predicts and describes changes in particle motion, temperature, and state of a pure substance when thermal energy is added or removed. Writing: Support claims with logical reasoning and relevant, accurate data and evidence that demonstrate an understanding of the topic or text, using credible sources. 2015-2016 6 Curriculum and Instruction – Office of Science Middle School Science: Map for Grade 8 First Nine Weeks possible sources of bias and error, alternative explanations, and questions for further exploration. makeup of the atmosphere illustrates a mixture of gases. Reading: Follow precisely a multistep procedure when carrying out experiments, taking measurements, or performing technical tasks. Writing: Use precise language and domain- specific vocabulary to inform about or explain the topic. Unit 1.3 Elements, Compounds, and Mixtures, 3 weeks Academic vocabulary: atom, atomic number, element GLE 0807.9.1: Understand that all matter is made up of atoms. GLE 0807.9.4: Distinguish between elements, compounds, and mixtures GLE0807.9.6: Use the periodic table of elements to determine the characteristics of an element. GLE 0807.Inq.1: Design and conduct open-ended scientific investigations. Design and conduct an investigation to discriminate between types of mixtures. GLE 0807.Inq.3 Synthesize information to determine cause and effect relationships between evidence and explanations. Demonstrate that the properties of a compound are different from the properties of its component elements. Differentiate between elements and compounds using chemical symbols and formulas. Relate the word “element” to “compound” and “atom” to Tennessee Holt Science and Technology TE, Chapter 9, Section 1: Elements and Section 2: Compounds and Section 3: Mixtures p. 232-249. Chapter12, Section 1: Electrons and Chemical Bonding p. 314317. Glencoe Tennessee Science 8; Chapter 7 p. 190-213; Chapter 8 p.220-249; Chapter 11 p. 306320. STCMS Properties of Matter TG: NGSS Practice 7: Engaging in argument from evidence NGSS Practice 8: Obtaining, evaluating, and communicating information NGSS Practice 1: Asking questions and defining problems NGSS Practice 3: Planning and carrying out investigations Writing: Support claims with 2015-2016 7 Curriculum and Instruction – Office of Science Middle School Science: Map for Grade 8 First Nine Weeks “molecule.” Lessons 11, 15-17, and 20, p. 125-134, 161-192, and 227-240. Demonstrate a variety of methods to separate the components of compounds and mixtures. Justify the classification of mixtures and compounds by their properties. logical reasoning and relevant, accurate data and evidence that demonstrate an understanding of the topic or text, using credible sources. Reading: Follow precisely a multistep procedure when carrying out experiments, taking measurements, or performing technical tasks. Writing: Use precise language and domain- specific vocabulary to inform about or explain the topic. TOOLBOX Unit 1.1 Properties of Matter, 3 weeks Unit 1.1 Properties of Matter American Chemical Society (ACS) Lesson about Density: Students will be able to calculate the density of different cubes and use these values to identify the substance each cube is made of. Students will be able to explain that the size, mass, and arrangement of the atoms or molecules of a substance determine density. http://www.middleschoolchemistry.com/lessonplans/chapter3/lesson1 Plans ACS lesson about density of solids: Students will investigate a wax candle and a piece of clay to understand why the candle floats and the clay sinks even though the candle is heavier than the piece of clay. Students will discover that it is not the weight of the object, but its density compared to the density of water, that determines whether an object will sink or float in water. http://www.middleschoolchemistry.com/lessonplans/chapter3/lesson4 ACS lesson about density of liquids: Students will observe three household liquids stacked on each other and conclude that their densities must be different. They will predict the relative densities of the liquids and then measure their volume and mass to see if their calculations match their observations and predictions. 2015-2016 8 Curriculum and Instruction – Office of Science Middle School Science: Map for Grade 8 First Nine Weeks http://www.middleschoolchemistry.com/lessonplans/chapter3/lesson5 Teacher’s Lesson plans, labs, notes, homework. http://betterlesson.com/community/unit/15360/matter You will need to create an account. Video and printed material for all Chemical and Physical Science http://www.gpb.org/chemistry-physics/students/all Unit 1.1 Properties of Matter Background for Teachers Unit 1.1 Properties of Matter Other Resources If you've been assigned to teach principles of chemistry, but haven't studied the topic recently (or perhaps ever!) consult the following website and select the topics for review: http://www.inquiryinaction.org/chemistryreview/ Good general website for background information: http://phet.colorado.edu/ Chemistry related middle school science resources: http://www.science-class.net/archive/science-class/Chemistry/Chemistry.htm and http://www.middleschoolscience.com/chemistry.htm Online Density Quiz. Students determine the units, tools, and how to calculate density. http://www.quia.com/quiz/1145135.html?AP_rand=1573855015 An online quiz requires students to classify each given change as physical or chemical. There are no tutorials to assist struggling learners. http://www.quia.com/quiz/303980.html This website gives definitions, examples and general information about matter. http://www.chem4kids.com/files/matter_intro.htmlhttp://www.chem4kids.com/files/matter_intro.html Information about Physical and Chemical changes: http://www.chem4kids.com/files/matter_chemphys.html Information about atoms: http://www.chem4kids.com/files/atom_structure.html Other Resources (cont’d) A collection of websites for answering the question "How can physical properties be used to identify matter?" http://www.scilinks.org/Harcourt_Hsp/HspStudentRetrieve.aspx?Code=HSP513 2015-2016 9 Curriculum and Instruction – Office of Science Middle School Science: Map for Grade 8 First Nine Weeks Unit 1.2 Phases of Matter, 2 weeks Unit 1.2 Phases of Matter Plans American Chemical Society (ACS) lesson plan for melting: Students will see a small piece of ice melt on an aluminum surface. Students will explain the energy transfer and molecular motion which cause the change in state from a solid to a liquid. Students will see and discuss an animation of ice melting and compare the state changes of water to the state changes of other substances. They will also investigate sublimation of dry ice through a teacher demonstration, or video if dry ice is not readily available. http://www.middleschoolchemistry.com/lessonplans/chapter2/lesson5 ACS lesson about solids: Students will see a demonstration with a metal ball and ring showing that heat causes atoms to spread a little further apart. They will also see that cooling a solid causes the atoms to get a little closer together. The same rules they discovered about liquids also apply to solids. http://www.middleschoolchemistry.com/lessonplans/chapter1/lesson4 ACS Lesson about gases: This lesson focuses on molecular motion in gases. Students compare the mass of a basketball when it is deflated and after it has been inflated. The inflated ball has the greater mass so students can conclude that gas is matter because it has mass and takes up space. Then students consider how heating and cooling affect molecular motion in gases. They dip the mouth of a bottle in detergent solution and observe a bubble growing and shrinking when the bottle is warmed and cooled. Students will learn that the attractions between gas molecules are so minimal that attractions can’t be used to explain the behavior of gases like it can for liquids and solids. http://www.middleschoolchemistry.com/lessonplans/chapter1/lesson5 Unit 1.2 Phases of Matter Other Resources A great refresher about the phases of matter: http://www.chem4kids.com/files/matter_states.html Gas Law and Pressure Labs: http://www.chymist.com/Exps%20with%20a%20140%20mL%20syringe.pdf 2015-2016 10 Curriculum and Instruction – Office of Science Middle School Science: Map for Grade 8 First Nine Weeks Unit 1.3 Elements, Compounds, and Mixtures, 3 weeks Unit 1.3 Elements, Compounds, and Mixtures Plans Unit 1.3 Elements, Compounds, and Mixtures Great lesson for grouping elements. Easy transition to the Periodic Table of Elements: http://betterlesson.com/lesson/622633/common-groups-of-elements Teacher’s Lesson plans, labs, notes, homework. http://betterlesson.com/community/unit/15822/atoms-and-the-periodic-table You will need to create an account. Explore chromatography with your students by making a string of "light bulbs!" Students cut light bulb shapes out of coffee filters or filter paper and use watersoluble markers, pipe cleaners, and water to create a colorful display. http://sciencespot.net/Pages/classchem.html#Anchorhalls 2015-2016 11