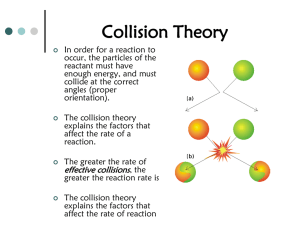
Chemistry: Reaction Rates & Equilibrium - Standard Analysis
advertisement

Belding Area Schools Standard Analysis Unit 4 Chemistry: Reaction Rates and Equilibrium STRAND: C STANDARD: See HSCE SUBJECT: Chem. B Unit 4 I can compare the entropy of solids, liquids and gases. (C2.2e) I can draw and analyze a diagram to show the activation energy for an exothermic reaction that is very slow at room temperature. (C2.3b) I can predict how the rate of a chemical reaction will be influenced by changes in concentration, and temperature, pressure. (C5.r1a) I can explain how the rate of a reaction will depend on concentration, temperature, pressure, and nature of reactant. (C5.r1b) I can describe equilibrium shifts in a chemical system caused by changing conditions (Le Chatelier’s Principle). (C5.3a) I can predict shifts in a chemical system caused by changing conditions (Le Chatelier’s Principle). (C5.3b) I can predict the extent reactants are converted to products using the value of the equilibrium constant. (C5.3c) I can predict if a chemical reaction is spontaneous given enthalpy and entropy changes for the reaction using Gibb’s Free Energy. (C3.4e) I can explain why some endothermic reactions are spontaneous at room temperature. (C3.4f) I can explain why gases are less sloluble in warm water than cold water. (C3.4g) Enduring Understanding(s): Changing conditions will cause shifts in the equilibrium of a chemical reaction. In a closed system, many reactions reach equilibrium. The concentration of a solution affects the reaction rate of chemical reactions. Essential Questions(s): How do changing conditions affect the equilibrium of a chemical system? How does the concentration of a solution affect the rate of chemical reactions? Vocabulary: Heat of solution Revised March, 2014 Belding Area Schools Standard Analysis Le Chatelier’s Principle Reaction Rate Entropy HSCE (High School Content Expectations) See above Information/Rules/Procedures/Resources/Assessments Instructional Strategies for all students: See instructors Moodle page for daily lesson plans, activities, labs, power points, homework, and tutorials. Differentiated Instruction for at-risk students: After school study sessions. Review Guides Tutorials on Moodle Retesting opportunity on all assessments Co-taught classes available Assessments: On staff share drive. Revised March, 2014