Content
advertisement

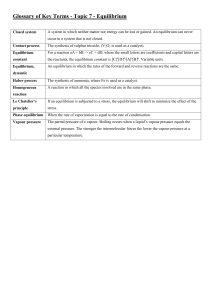
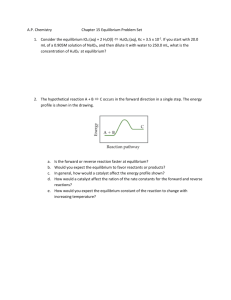
Title: Chemistry Digital Agenda Date: 3/1-2/2012 Content: o Teacher: Introduction to the Unit/Inquiry exploration of rates of chemical reactions Mr. Jeff Astor Date: 3/1-2/2012 Subject/Course: Chemistry Grade: 9-10 Do Now: Catalyst is the Terminology Takedown CA Standard(s): Standard(s) from Instructional Guide 8a Students know the rate of reaction is the decrease in concentration of reactants or the increase in concentration of products with time. 8b Students know how reaction rates depend on such factors as concentration, temperature, and pressure. 8c Students know the role a catalyst plays in increasing the reaction rate. 8d Students know the definition and role of activation energy in a chemical reaction. 9a Students know how to use Le Châtelier’s principle to predict the effect of changes in concentration, temperature, and pressure. 9b Students know equilibrium is established when forward and reverse reaction rates are equal. Learning Objective (s): SWBAT… LT: 8.5 – I can describe what equilibrium is and relate it to the rate of the forward and reverse reactions. 8.6 – I can define Le Chatelier’s principle and use this principle to predict how concentration, temperature, and pressure will impact a given equilibrium. Focus Question: What is the process by which food decomposes? Assessment: Summative Test: Temperature and Thermodynamics on March 14 Benchmark III-March 26-March 30 Daily Agenda: (check off each assignment after completion) o Catalyst Involves the Terminology Takedown which will incorporate words involving reaction kinetics and equilibrium concentrations. o o Hook – Paper ball fight JTPS – Why did the reaction reach equilibrium? 10 o o o Direct Instruction Station – Students will watch a video where Mr. Astor will go over a power point explaining what equilibrium is, how it is established, and --how it is related to rates of reactions. He will also discuss how it is effected by changing what is in the reaction, and relate that to Le Chatelier’s Principle. Collaborative Work Station – Students will create and form a collaborative poster with the main ideas of equilibrium established concretely on the poster. There will be a rubric and guidelines for each student, and the students at the collaborative station will be assessing each other on the effectiveness of their poster creation. Independent Work Station – Students will log on to Ed Elements and complete the section on equilibrium and reaction rates, as well as reversible reactions. Assessment – Defining equilibrium, what it is, and how it is related to rates of reaction – Exit Slip o Practice: Finish Practice 8.3 – Equilibrium Equations How will students reflect on what they’ve learned? Review of lab activities Review of exit slips