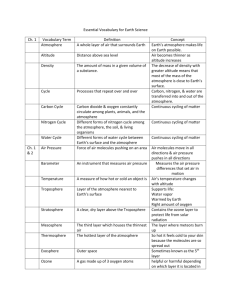
Properties of Elements_Card Size
advertisement

Element: A Physical Properties: colorless monatomic gas; density less than atmosphere; IP = 24.6 eV; MP = -272°C; BP = -269°C Element: B Physical Properties: colorless monatomic gas; density similar to that of the atmosphere; IP = 21.6 eV; MP = -249°C; BP = -246°C Chemical Properties: not reactive Chemical Properties: not reactive Element: C Physical Properties: colorless monatomic gas; density greater than that of the atmosphere; IP = 15.8 eV; MP = -189°C; BP = -186°C Chemical Properties: not reactive Element: E Physical Properties: conducts electricity and heat in its brittle, black, solid form; not conductive in its very hard, crystalline form; IP = 11.3 eV; MP = 3652°C Chemical Properties: reacts with oxygen to form monoxides and dioxides, forms tetra halides. Element: D Physical Properties: IP = 10.5 eV; MP = 44°C; BP = 2800C Chemical Properties: forms many different oxides Element: F Physical Properties: pale yellow diatomic gas; IP = 17.4 eV; MP = -220°C; BP = -188°C Chemical Properties: forms binary compounds with most metals and all semiconductors. Element: G Element: H Physical Properties: colorless diatomic gas; density less than that of the atmosphere; IP = 13.6 eV; MP = -259°C; BP = -253°C Physical Properties: greenish colored diatomic gas; IP = 13.0 eV; MP = -101°C; BP = -35°C Chemical Properties: reacts violently with oxygen. Chemical Properties: forms binary compounds with most metals and all semiconductors. Element: I Physical Properties: colorless diatomic gas; not attracted to magnet in its liquid or solid form; similar in density to the atmosphere; IP = 14.5 eV; MP = -210°C; BP = -196°C Chemical Properties: causes glowing splint to go out, forms many different oxides. Element: K Physical Properties: IP = 6.0 eV; MP = 660°C; BP = 2467oC Chemical Properties: forms trihalides Element: J Physical Properties: IP = 9.3 eV; MP = 1278°C; BP = 29700C Chemical Properties: forms a monoxide when reacted with oxygen Element: L Physical Properties: yellow solid; poor conductor of heat and electricity; IP = 10.4 eV; MP = 113°C; BP = 445°C Chemical Properties: reacts with oxygen, forms a dihydrogen compound. Element: M Physical Properties: colorless gas; attracted to a magnet in its liquid and solid form; density similar to that of the atmosphere; IP = 13.6 eV; MP = -218°C; BP = -183°C Chemical Properties: causes glowing splint to burst into flame, causes glowing steel wool to burst into flame, forms an orange compound when reacted with iron, forms a dihydrogen compound. Element: O Element: N Physical Properties: IP = 8.2 eV; MP = 1410°C; BP = 2355°C; semiconductor Chemical Properties: forms tetrahalides, forms dioxides Element: P Physical Properties: metallic finish; malleable; conducts electricity; conducts heat; IP = 7.7 eV; MP = 650°C; BP = 10900C Physical Properties: metallic finish; malleable; IP = 5.1 eV; MP = 98°C; BP = 883°C Chemical Properties: burns brightly in presence of oxygen to form a white powder, reacts with acid to form hydrogen gas, forms a monoxide when burned with oxygen. Chemical Properties: reacts quickly with the atmosphere, readily forms ions in water. Element: Q Physical Properties: metallic finish; malleable; IP = 5.4 eV; MP = 181°C; BP = 1342°C Chemical Properties: reacts quickly with the atmosphere, readily forms ions in water. Element: R Physical Properties: IP = 8.3 eV; MP = 2079°C; BP = 2550°C; semiconductor Chemical Properties: forms trihalides Element: A Element: A Physical Properties: Physical Properties: Chemical Properties: Chemical Properties: Element: A Element: A Physical Properties: Physical Properties: Chemical Properties: Chemical Properties: Element: A Element: A Physical Properties: Physical Properties: Chemical Properties: Chemical Properties: