supercritical fluid extraction of essential oils
advertisement

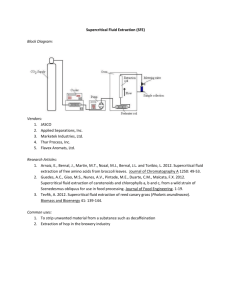
Session C11 6124 Disclaimer — This paper partially fulfills a writing requirement for first year (freshman) engineering students at the University of Pittsburgh Swanson School of Engineering. This paper is a student, not a professional, paper. This paper is based on publicly available information and may not be provide complete analyses of all relevant data. If this paper is used for any purpose other than these authors’ partial fulfillment of a writing requirement for first year (freshman) engineering students at the University of Pittsburgh Swanson School of Engineering, the user does so at his or her own risk. SUPERCRITICAL FLUID EXTRACTION OF ESSENTIAL OILS Madeleine Hamrick, mch59@pitt.edu, Vidic 2:00, Julie Steranka, jms502@pitt.edu, Mahboobin 10:00 Revised Proposal — Supercritical fluid extraction, or SFE, is currently the main method used for extracting essential oils. The SFE process has received much attention in recent years as it offers many more advantages over alternative methods, such as organic solvent extraction and steam distillation, for recovering essential oils from plants. Supercritical fluids for the extraction of essential oils makes the process faster, easier, and more efficient than previously used methods. A supercritical fluid is a substance at a temperature and pressure that is above its critical point therefore allowing a substance’s liquid and vapor phase to coexist. The fluids can effuse through solids like a gas and can dissolve materials like a liquid [1]. This unique characteristic of supercritical fluids makes them ideal for extracting oils from dense plant material. Supercritical fluid extraction is a process of splitting one constituent from another using supercritical fluid as the extracting solvent. The extraction of essential oils using supercritical fluids works by allowing a supercritical fluid to diffuse into plant materials thus dissolving the material to be extracted, in this case, the essential oil. The dissolved material can then be separated from the plant and cooled in order to isolate the essential oils [2]. The extraction of essential oils has become more dependent on the use of SFE techniques. This process is significant to engineers because essential oils play a main role in pharmaceutical, food and fragrance industries. For example, essential oils are key ingredients in medicine, perfumes, cosmetics, and foods [3]. Chien Qingyong Lang mentions, “With increasing public interest in natural products, SFE may become a standard extraction technique for studying herbal food and agricultural samples” [4]. In addition, essential oils have many complex components whose proportions must be maintained during extraction. Supercritical fluids are an ideal option because they not only speed up the extraction process, but also keep the proportions intact [3]. The conference paper will detail what essential oils and supercritical fluids are, what the process of supercritical fluid extraction is, and how this process is now used to extract these oils. It will continue by highlighting the importance, ethical issues surrounding this topic, and improvements that could be made. Online journal articles mentioned in the “Sources Consulted” section will provide University of Pittsburgh Swanson School of Engineering 1 29 January 2016 most of the information. Diagrams and equations will also be used to clarify this process and its significance. REFERENCES [1] V. Krukonis. (1998). “Supercritical Fluids: Their Proliferation in the Pharma Industry.” Phasex Corporation. (online article). http://www.phasex4scf.com/supercriticalfluids-applications [2] S. Hawthorn. (1990). “Analytical –Scale Supercritical Fluid Extraction.” Analytical Chemistry. (online article). http://pubs.acs.org/doi/abs/10.1021/ac00210a722 [3] S. Pourmortazavi, S. Hajimirsadeghi. (2007). “Supercritical fluid extraction in plant essential and volatile oil analysis.” A Journal of Chromatography.. (online article). DOI:10.1016/j.chroma.2007.06.021 [4] Q. Lang. (2001). “Supercritical fluid extraction in herbal and natural product studies — a practical review.” Talanta. DOI:10.1016/S0039-9140(00)00557-9 ANNOTATED BIBLIOGRAPHY K. Ara, M. Jowkarderis, F. Raofie. (2015). “Optimization of supercritical fluid extraction of essential oils and fatty acids from flixweed (Descurainia Sophia L.) seed using response surface methodology and central composite design.” SpringerLink. DOI: 10.1007/s13197-014-1353-3 This article, from the Journal of Food Science and Technology explains in depth the mechanism and procedure for extracting essential oils using supercritical fluid. It mentions the many uses of essential oils after they are isolated. This article also provides data from experimentation comparing the yields from different extraction methods. This article will provide us with factual data and results to prove SFE as an ideal process of extraction. R. Da Silva. (2016). “Supercritical fluid extraction of bioactive compounds.” TrAC Trends in Analytical Chemistry. DOI:10.1016/j.trac.2015.11.013. This article, from a scientific journal about analytical chemistry explains how supercritical fluid extraction is an ideal, alternative green technology for extraction methods. It offers an overview of the application of the process. The Madeleine Hamrick Julie Steranka S. Pourmortazavi, S. Hajimirsadeghi. (2007). “Supercritical fluid extraction in plant essential and volatile oil analysis.” A Journal of Chromatography.. (online article). DOI:10.1016/j.chroma.2007.06.021 This article, from a peer reviewed scientific journal focusing on chromatography, discusses the factors that affect SFE. Pressure, quantity, porosity of the plant, and the solubility of the supercritical fluid can be modified to extract essential oils from plants. The essential oils are complex compounds that must stay intact during the extraction. We will use this article to highlight how the SFE process can be modified to extract different oils. article investigates improvements that can be made on the extraction efficiency and techniques. We will use this source to detail the improvements that can be made to the SFE process and its prevalence over other methods. S. Hawthorn. (1990). “Analytical –Scale Supercritical Fluid Extraction.” Analytical Chemistry. (online article). http://pubs.acs.org/doi/abs/10.1021/ac00210a722 This article, from the American Chemical Society journal explains previous methods of extraction using liquid solvents and their drawbacks. The article highlights supercritical fluid as an alternative method of extraction. The article describes current techniques, applications, capabilities, and limitations using supercritical fluids in extraction and recovering organic materials. Information from this article will help clarify the significance of using the supercritical fluid method of extraction over other processes. M. Zougagh, M. Valcarcel. (2004). “Supercritical fluid extraction: a critical review of its analytical usefulness.” TrAC Trends in Analytical Chemistry. DOI:10.1016/S01659936(04)00524-2 This article, from a scientific journal about analytical chemistry offers a critical review of the usefulness of the supercritical fluid extraction process. The article examines the evolution of the process in the past thirty years. This article provides reasons for its rare implementations and proposes ways to overcome the defects behind them. This article will be used to describe fixes that can be made to enhance the SFE process. M.Herrero, J. Mendiola, A. Cifuentes, E. Ibáñez. (2010). “Supercritical fluid extraction: Recent advances and applications.” A Journal of Chromatography. (online article). DOI:10.1016/j.chroma.2009.12.019 This article, from a peer reviewed scientific journal focusing on chromatography, details how the use of supercritical fluids is increasing in industries. The main compound used is carbon dioxide, because it is cheap, environmentally friendly, and considered safe by the FDA. It is becoming more extensively used in food and natural products. This information will help show the rapidly increasing applications of this process. V. Krukonis. (1998). “Supercritical Fluids: Their Proliferation in the Pharma Industry.” Phasex Corporation. (website). http://www.phasex4scf.com/supercritical-fluidsapplications This article, from a company’s website that deals with the application of supercritical fluids, explains the benefits of using supercritical fluids in the extraction process. Due to this its solubility property, there is less residue and higher yields after the process when compared to previously used methods. We will use this information to describe the benefits of using highly soluble supercritical fluids. Q. Lang, C.Wei. (2001). “Supercritical fluid extraction in herbal and natural product studies — a practical review.” Talanta. DOI:10.1016/S0039-9140(00)00557-9 This article, from a peer reviewed international journal about analytical chemistry, describes the benefits of using SFE such as high viscosity and easy manipulations. It also describes how the process is performed and can be modified to be selective. This article will be used to explain the process of SFE and how it can be altered to obtain certain essential oils. 2






![Heat transfer mechanisms Nucleation at high subcritical pressures [1]](http://s3.studylib.net/store/data/006613018_1-484ac98340bdf87d83d3defecfde6c98-300x300.png)