Heat transfer mechanisms Nucleation at high subcritical pressures [1]
advertisement
![Heat transfer mechanisms Nucleation at high subcritical pressures [1]](http://s3.studylib.net/store/data/006613018_1-484ac98340bdf87d83d3defecfde6c98-768x994.png)
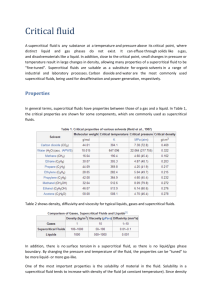
Chapter 3 Heat transfer mechanisms and regimes 1. Heat transfer mechanisms 1.1 Nucleation at high subcritical pressures [1] Nucleation at a boiling site of a given size becomes easier as the pressure of a boiling liquid increases. The decrease of surface tension means that a smaller difference between vapour and liquid pressure is required in order to make a nucleus grow; moreover, a given pressure difference can be achieved with a smaller superheat as the pressure level increases. For a given superheat, therefore, it will be possible to activate smaller nuclei at higher pressures. In addition to this effect, there is the possibility of spontaneous nucleation occurring as a result of thermal fluctuations in a superheated liquid at a surface as p/pc approaches unity. Indeed, it may be that the very "quiet" transition to film boiling that occurs at high pressure may be a consequence of spontaneous nucleation. 1.2 Pool boiling [1] The effect of pressure on nucleate boiling is well established: as the pressure is raised towards the critical pressure the heat transfer coefficient increases, and the limiting heat flux for transition from nucleate to film boiling first increases, reaches a maximum at a value of p/pc of about 0.35, and then decreases. Pool boiling experiments at high pressures show very clearly how the range of conditions under which nucleate boiling occurs is contracted by transition to film boiling. Following transition to film boiling at pressures close to the critical value, a highly regular flow of vapour from the heated surface is observed. At p/pc ≈ 0.9 uniformly spaced columns of bubbles are observed to rise from a heated wire. As the critical pressure is approached these columns change into "tubes" of vapour, again with very regular spacing, and finally change further into what appears to be a sheet of vapour rising from the wire. 1.3 Flow boiling [1] As indicated earlier, the general picture of supercritical pressure forced convection has become comparatively clear. In contrast, very few detailed investigations have been made at high subcritical pressures. Figure 1 shows data for water flowing upwards in a 20-mm diameter pipe for pressures extending from subcritical to slightly supercritical, taken from the work of Herkenrath et al. It is of interest to compare the subcritical curves, each of which involves a local heat transfer crisis, with the curve for supercritical pressure, which does not. Note that the conditions are such that buoyancy induced temperature peaks would not be expected in the case of the supercritical data. Figure 1: Wall-temperature versus bulk-enthalpy for water flowing upwards in a 20-mm diameter pipe for pressures extending from subcritical to slightly supercritical (Herkenrath et al.) It will be seen from Figure 1 that as the pressure is increased the position of the heat transfer crisis shifts towards the region of lower quality; the magnitude of the wall temperature step at first decreases, then increases and finally decreases as the critical pressure is approached. Furthermore, the shape changes from a sharp to a more gradual increase in wall temperature. Some data obtained by the present author and co-workers using carbon dioxide at a reduced pressure, p/pc = 0.99, for both upward and downward flow showed an increase in wall temperature following transition which was rather gradual (the curve for p = 215 bar in Figure 1) and that in upward flow peaks develop due to buoyancy influence as they do at supercritical pressure. It was also apparent that at high subcritical pressures downward flow was advantageous, just as at supercritical pressures: transition occurred later in downward flow and the consequences were less severe. Experiments with carbon dioxide at lower pressure show a sharp transition rather similar to those illustrated in Figure 1. The beneficial consequences of buoyancy in downward flow do not appear to persist as the pressure is dropped further, where the above mentioned trend appears to be reversed. [1] J. D. Jackson, „Supercritical heat transfer,” [Online]. Available: http://www.thermopedia.com/content/1173/. [Geopend 12 11 2012]. [2] Kyoung-Ho Kang, Soon-Heung Chang. Experimental study on the heat transfer characteristics during the pressure transients under supercritical pressures, International Journal of Heat and Mass Transfer 52 (2009) 4946–4955. [3] W.B. Hall, Heat Transfer near the Critical Point, Advances in Heat Transfer, Volume 7, 1971 , p.186. [4] Cheng X, Schulenberg T; Heat transfer at supercritical pressure literature review and application to an HPLWR, Forschungszentrum Karlsruhe, 2001. [5] Groeneveld, DC et al; A general method of predicting critical heat flux in advanced water-cooled reactors. Proc. of NURETH-9, October 1999, San Francisco. [6] K. Yamagata, K. Nishikawa, S. Hasegawa, T. Fujii, S. Yoshida, Forced convection heat transfer to supercritical water flowing in tubes, Int. J. Heat Mass Transfer, Vol.15 (1972), pp.2575-2593. [7] S. Koshizuka, N. Takano, Y. Oka, Numerical analysis of deterioration phenomena in heat transfer to supercritical water, Int. J. Heat Mass Transfer, Vol.38, No.16 (1995), pp.3077-3084. [8] M.A. Styrikovich, T.K. Margulova, Z.L. Miropolskiy, Problem in the development of designs of supercritical boilers, Teploenergetika, Vol.14, No.6, 1967, pp.4-7. [9] Jackson, J D and Hall, W B, Influences of buoyancy on heat transfer to fluids flowing in vertical tubes under turbulent conditions, Turbulent Forced Convection in Channels and Rod Bundles, 1979, Vol2., 613-640, published by Hemisphere Publishing Corporat. [10] H. Ogata, S. Sato, Measurement of forced convection heat transfer to supercritical helium, Proc. of the 4th. Int. Cryogenic Engineering Conference, Eindhoven, Netherlands, may 24-26, 1972, pp.291-294. [11] B.S. Petuhkov, V.A. Kurganov, Heat transfer and flow resistance un the turbulent pipe flow of a fluid with near-critical state parameters, Teplofizika Vysokikh Temperature, Vol.21, No.1, pp.92100, Jan. 1983.