CLASS COPY
Do not write on this handout.
Biology
Enzyme Lab – Part A: The effect of organic and inorganic catalysts on the rate of hydrogen peroxide
decomposition.
For this lab: You need to include the title, objective, pre-lab questions, hypothesis, summarized procedure,
TAPED IN data table, graph, analysis questions, and conclusion.
Objective: Measure the effect of catalysts (enzymes) on a chemical reaction in a controlled experiment.
Introduction:
In each individual cell of a human there are many chemical reactions taking place, performing the necessary
functions for being a large, complex, multicellular organism. This is relatively easy to understand. How do
these reactions occur? This is not so easy to understand. Chemical reactions involve the breaking and
reforming of chemical bonds between molecules (substrate(s) of the reaction), which are transformed into
different molecules (product(s) of the reaction). Chemical reactions can occur spontaneously (without added
energy or intervention), and indeed many of the chemical reactions necessary for life processes are
spontaneous; some however, are not.
What would happen to your cells if they made a poisonous chemical? You might think that they would die. In
fact, your cells are always making poisonous chemicals. They do not die because your cells use enzymes to
break down these poisonous chemicals into harmless substances. Enzymes are proteins that speed up the rate
of reactions that would otherwise happen more slowly. The enzyme is not altered by the reaction. You have
hundreds of different enzymes in each of your cells.
Each of these enzymes is responsible for one particular reaction that occurs in the cell. In this lab, you will
study an enzyme that is found in the cells of many living tissues. The name of the enzyme is catalase (KAT-uhLAYSE). It speeds up a reaction which breaks down hydrogen peroxide, a toxic chemical, into 2 harmless
substances--water and oxygen.
The reaction is: 2 H2O2 + enzyme ----> 2 H2O + O2
This reaction is important to cells because hydrogen peroxide (H2O2) is produced as a byproduct of many
normal cellular reactions. If the cells did not break down the hydrogen peroxide, they would be poisoned and
die. In this lab, you will study the catalase found in liver cells. You will be using chicken or beef liver. It might
seem strange to use dead cells to study the function of enzymes. This is possible because when a cell dies, the
enzymes remain intact and active for several weeks, as long as the tissue is kept refrigerated.
PRELAB Questions:
1. What is an enzyme?
2. What to enzymes do?
3. Define catalyst.
4. Complete the following: All enzymes are _________________, but not all _______________are
enzymes.
5. Write the chemical equation above. Circle the reactant(s) in the equation above. Put a square around
the product(s) in the equation above.
6. What is our substrate in this lab?
7. What is our enzyme in this lab?
8. What indication will we have that the enzyme is working? HINT: Look at the products, what would
having these together LOOK like?
Experimental Design Diagram
In this experiment you will compare the effect of the type of catalys used on the speed of a reaction.
All scientific experiments need to have an independent variable and a dependent variable. Additionally, one
must always consider what you will use as a control and what other variables you need to hold constant. In
this experiment we will have the following:
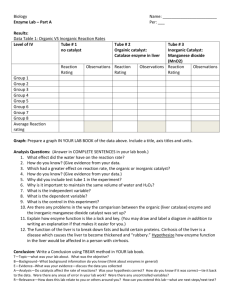
Independent Variable: Type of catalyst added
Level of IV
No catalyst
Organic catalyst:
Inorganic Catalyst:
Catalase enzyme in liver
Manganese dioxide
(MnO2)
Number of
1 per group
1 per group
1 per group
repeated trials for
each level
Dependent Variable: Speed of Reaction
Throughout this investigation you will estimate the rate of the reaction (how rapidly the solution bubbles) on a
scale of 0-5 (0=no reaction, 1=slow, ..... 5= very fast).
Control: (What you compare your results to) Test tube # 1 with no catalyst added.
Constants: (Factors you kept the same because they could affect the rate of reaction)
- Amount of hydrogen peroxide in each test tube
- Time between adding catalyst and measuring rate of reaction
- Are there others we ought to consider?
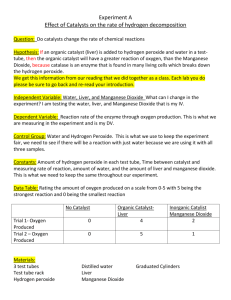
Materials:
3 test tubes
1 test tube rack
Hydrogen peroxide (H2O2)
Distilled water
Liver
Manganese dioxide(MnO2)
Graduated cylinder(s)
Procedure:
1. SAFETY: Wear goggles throughout the lab.
2. Obtain three test tubes and label them 1, 2, and 3.
3. VERY IMPORTANT: DO NOT ADD THE H2O2 to tubes #2 and 3 until you are running the experiment for
that test tube.
4. Prepare the contents of each test tube as outlined below ONE AT A TIME:
Test tube #1 – 10 mL Water and 10 mL hydrogen peroxide (H2O2)
Test tube #2 – 10 mL Water and 10 mL H2O2 and small piece of liver
Test tube #3 – 10 mL Water and 10 mL H2O2 and small (pea size) amount of MnO2
5. Record your observations about each tube in addition to rating the reaction (how rapidly the solution
bubbles - on a scale of 0-5 where 0=no reaction, 1=slow, ..... 5= very fast)
6. Clean up as instructed.
7. Record the data from the other classroom groups.
8. Calculate the average the reaction ratings for each test tube (sum the ratings and divide by the number
of entries).
9. Create a graph of your results.
10. Answer the analysis questions.
 0
0