Chemistry Assignments Week of 11.19.12
advertisement

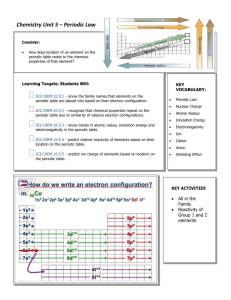
Chemistry Assignments Week of 11.19.12 M 5 T 6 Chem 1 Per 1 AP Chem P 3 Chem II P 6/7 Chem IH P 9/10 Chem IH Per 12 Define the mole, avogadro’s number and molar mass. Use Molar Mass as a conversion factor. Determine the pressure in a mixture of gases by applying Dalton’s Law of Partial Pressure. Determine the pressure of a mixture of gases at equilibrium. Describe the steps in preparing a solution of a given molar concentration from a concentrated solution. Write longhand electron configurations for atoms, using the periodic table. Write longhand electron configurations for atoms, using the periodic table. Mass Mole WS #s 1,2,3 a,c,e,g Use Molar Mass and Avogadro’s number to convert from grams to moles and atoms Mass Mole Conversion Charts in Notes packet. W WS: Lecture 19 #s 1-11 WS Solutions # 8993 Practice solving solution reaction and dilution problems. Textbook problems Chapter 4#s 60,62,66,70,74,86, 88b,106 Note packet 3 page 1 Using a periodic table, write electron configurations for elements that are: 1. Exceptions to the aufbau order of filling 2. Abbreviations 3. ions. Note packet 3 pages 2,3 Note packet 3 page 1 Using a periodic table, write electron configurations for elements that are: 1. Exceptions to the aufbau order of filling 2. Abbreviations 3. ions. Note packet 3 pages 2,3 Chemistry Assignments Week of 11.19.12 TH F Week of 11.19.12