ABS worksheet 1 answer
advertisement

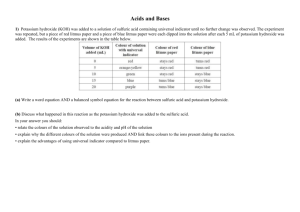
Hwa Chong Institution Acids, Bases and Salts (Worksheet 1) Name: __________________ ( ) Date: ___________________ Class: ___________________ Section A: Answer ‘True’ or ‘False’ to the following statements. Question All acidic solutions contain hydrogen ions. All bases produce hydroxide ions in water. Ethanoic acid is completely ionised in water. Copper(II) hydroxide is an alkali. Sulfuric acid is used in all batteries. A neutralization reaction results in water and hydrogen gas produced. The concentration of an acid tells you whether it completely ionises in water. Acids in dry organic solvents react with carbonates to give carbon dioxide. The catalyst used in Contact process is vanadium(V) oxide. Magnesium hydroxide is a strong alkali. Section B: Answer all the questions in this section in the brackets provided 1. Which of the following is true of all acids in aqueous solution? A. They liberate ammonia from ammonium salt. B. They give hydrogen with any metal. C. They liberate carbon dioxide from carbonates. D. They turn red litmus paper blue. ( C ) 2. Which of the following pairs will react to give a gas? A. Ammonium chloride and potassium hydroxide. B. Sodium carbonate and calcium hydroxide. C. Sodium hydroxide and hydrochloric acid. D. Potassium hydroxide and sulphuric acid. ( A ) T/F T F F F F F F F T F 3. Which of the following reactions is not a neutralization reaction? A. Ammonia and ethanoic acid. B. Magnesium oxide and hydrochloric acid. C. Sodium carbonate and hydrochloric acid. D. Potassium hydroxide and citric acid. ( C ) 4. In which of the following is dilute sulfuric acid not behaving as an acid? A. H2SO4 + 2NaOH Na2SO4 + 2H2O B. H2SO4 + CuO CuSO4 + H2O C. H2SO4 + BaCl2 BaSO4 + 2HCl D. H2SO4 + Mg MgSO4 + H2 ( C ) Section C: Answer all the questions in this section in the spaces provided 1. Aspirin is a weak monobasic acid that has low solubility in water. We can represent the chemical formula of aspirin as HA to simplify matters. a. What is meant by a “weak monobasic acid”? It is an acid that does not dissociate completely in water, and only releases a maximum of one hydrogen ion per molecule of the acid. b. Write the chemical formula of the salt formed from the reaction of aspirin and sodium carbonate. NaA. c. Hence, write down the chemical equation between aspirin and sodium carbonate. 2HA + Na2CO3 CO2 + 2NaA + H2O. d. The salt formed from aspirin and sodium carbonate is highly soluble in water. Addition of nitric acid to this salt causes aspirin to precipitate out again. Write the chemical equation, with state symbols, for the precipitation of asparin when nitric acid reacts with the salt of aspirin. NaA(aq) + HNO3(aq) NaNO3(aq) + HA(s) e. When a person takes the salt of aspirin, precipitation of aspirin takes place in the stomach. Why is this so? There is hydrochloric acid in the stomach which causes it to precipitate out. 2. Some metals react with acids. Name two other types of substances which also react with acids. Then write an equation for one example of each the two substances above. Metal carbonates/metal oxides/metal hydroxides. For e.g., Na2CO3 + 2HCl H2O + 2NaCl + CO2 For e.g., 2NaOH + H2SO4 2H2O + Na2SO4 3. It is said that under the effects of a bee’s sting, sodium bicarbonate, of NaHCO 3, can be used to counteract it. i) What can you deduce about the nature of a bee’s sting? A bee’s sting is acidic. ii) What gas do you think is evolved when you treat a bee’s sting with sodium bicarbonate? Carbon dioxide. iii) How would you test for the gas stated above? Pass the gas through a delivery tube into limewater. If a white precipitate is formed, carbon dioxide gas is present. 4. Weiwen did an experiment using silver carbonate and three different acids: hydrochloric acid, nitric acid and ethanoic acid. The concentrations of acids used were the same. To Weiwen’s surprise, after a brief effervescence, the reaction of silver carbonate and hydrochloric acid stopped whereas the reaction with nitric acid and ethanoic acid went to completion. a) Suggest a reason why the reaction of silver carbonate with hydrochloric acid stopped. The formation of the insoluble salt silver chloride will coat the silver carbonate, preventing further reaction with hydrochloric acid. b) Predict whether there was more effervescence for nitric acid or ethanoic acid. Nitric acid. c) Explain your answer in b). Nitric acid is a strong acid which ionises completely in water whereas ethanoic acid is a weak acid which only ionises partially. As a result, the higher concentration of H+ ions due to HNO3 will result in a faster reaction with nitric acid, hence more effervescence is observed with nitric acid.