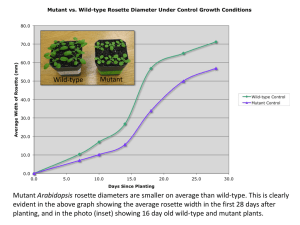
The Drosophila histone variant H2A
advertisement

The Drosophila histone variant H2A.V works in concert with HP1 to promote kinetochore-driven microtubule formation. Fiammetta Vernì and Giovanni Cenci Supplementary Figures Supplementary Figure 1. Segregation defects and spindle microtubule density in H2A.V mutants (A) DAPI staining of no-colchicine treated H2A.V (c,d) mutant neuroblasts that shows chromosomes and/or chromatids scattered along the major axis of the cell. Control metaphase and anaphase are shown in a) and b) respectively. Bar= 5m (B) Microtubule density from control (WT) and H2AV mutant cells. Note that mutant cells with misaligned chromosomes (S) show reduced density of chromosomal microtubules compared to either wild-type or mutant cells with normally aligned chromosomes (M). Centr MTs= Centrosomal Microtubules, Chrom MTs= Chromosome Microtubules (* p<0.01, t Student's Test) Supplementary Figure 2. Cyclin B localization upon loss of H2A.V. Cyclin B levels in H2A.V mutant cells with misaligned chromosomes (c,d) remain as high as in wild-type metaphases (a, b). Bar= 5 m. Supplementary Figure 3. H2A.V depletion does not affect localization of centromere/kinetochore proteins. Localization of centromere/kinetochore components, CID and Hec1 (red), CenpC and Mis12 (yellow) in H2A.V mutant PA cells is comparable to WT. Bar= 5 m. Supplementary Figure 4. Spindle defects and localization of cyclin B and centromere/ kinetochore antigens in HP1 mutant cells (A) Frequency of normal bipolar and irregular metaphase spindles in the 69B GAL 4>UAS HP1 RNAi mutant line. A total number of 140 cells have been examined. (B) A PA cell (top) in HP1 mutants that exhibits high level of Cyclin B. The bottom panel shows a characteristic HP1 mutant anaphase with chromatin bridges in which Cyclin B has been degraded. (C) Localization of centromere/kinetochore components, CID and Hec1 (red), and SAC protein BubR1 (yellow) in HP1 mutant PA cells; note the strong localization of BubR1 on PA, which indicates that in these cells the SAC is not satisfied. Bar= 5 m