Virtual Lab
advertisement

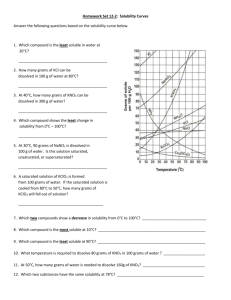
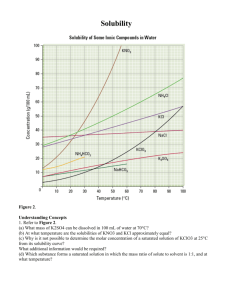
Name: _______________________________ Date: ________________ Solubility Virtual Lab Objective: Today you will explore the effect of temperature and polarity on solubility of salt in water. Hypothesis: Create a hypothesis about how either polarity or temperature affect solubility. Procedure: 1. Go onto the Puffin App and then go to Mrs. Simon’s webpage. 2. Once on her webpage, click on Helpful and Fun Links and then go to the Solubility Virtual Lab. 3. When on the website, pick a salt compound. Choose KCL. 4. After you choose, then find how many grams of that salt compound you can put in the cup of water at 0 degrees C, 20 degrees C, 60 degrees C and 100 degrees C until it becomes saturated. You can tell the solution is saturated when undissolved compound appears at the bottom of the beaker. 5. Record your data below. 6. Repeat steps 3-5 with NaNo3 7. Create a line graph of your results and then answer the questions. Virtual Lab: Temperature (Celsius) 0 20 60 Grams of KCl the solution can hold Grams of NaNO3 the solution can hold 100 Data Analysis: Create a line graph to represent the data collected in the virtual solubility lab. Be sure to label your x and y axis, give your graph a title, and include a key. “Temperature (C)” should be the x axis and “Amount Dissolved (g)” should be the y axis. 1. What effect does temperature have on solubility? __________________________________________________________ __________________________________________________________ __________________________________________________________ __________________________________________________________ 2. Which salt do you think is more polar and why? __________________________________________________________ __________________________________________________________ __________________________________________________________ __________________________________________________________ Conclusion: Write a conclusion for the virtual lab you just performed. You might have to research on the iPad to figure out WHY the lab results came out the way they did. ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________