student prac sample
advertisement

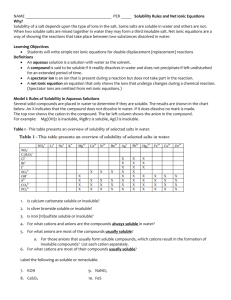
Solubility Prac Introduction: Solubility is when a particular substance can dissolve in water. The solute is the substance that dissolves in the solvent, (the solvent is usually of a larger amount) to form a solution which is two substances mixed together. When a substance, it is not soluble, when mixed with water, it will produce a precipitate which is a solid. The following table shows the solubility rules: Negative ions (anions) Positive ions (cations) Acetate, CH3COO– All All Li+, Cl– Solubility of compounds Soluble Na+, K+, Soluble Chloride, Bromide, Br– Iodide, I– All others Soluble Hydroxide, OH– Li+, Na+, K+, Rb+, NH4+, Sr2+, Ba2+ Soluble All others Low solubility Nitrate, PO43– Hg2+, NH4+ Ag+, NO3– Pb2+, Rb+, Cu+ Low solubility All Soluble Phosphate, Carbonate, CO32– Li+, Na+, K+, Rb+, NH4+ All others Low solubility Sulfate, SO42– Ca2+, Sr2+, Ba2+, Pb2+ Low solubility Soluble All others Sulfide, S2– Li+, Na+, K+, Soluble Rb+, NH4+, Be2+, Mg2+, Ca2+, Sr2+, Soluble Ba2+ All others Low solubility Aim: To predict, then test to determine which of the chosen ionic compounds are soluble in water. Materials: Lots of test tubes Test tube racks Water These ionic substances: Aluminium nitrate, Aluminium sulphate, Ammonium carbonate, Ammonium chloride, Ammonium sulphate, Barium chloride, Barium nitrate, Barium sulphate, Calcium carbonate, Calcium chloride, Calcium hydroxide, Calcium sulphate, Cobalt chloride, Copper II carbonate, Copper II oxide, Copper II sulphate, Iron II chloride, Iron III chloride, Iron III oxide, Lead chloride, Lead nitrate, Lithium chloride, Magnesium hydroxide, Magnesium nitrate, Magnesium oxide, Magnesium sulphate, Nickel sulphate, Potassium carbonate, Potassium chloride, Potassium dichromate, Potassium hydroxide, Potassium iodide, Potassium sulphate, Silver nitrate, Sodium carbonate, Sodium chloride, Sodium hydroxide, Sodium nitrate, Sodium sulphate, Strontium chloride. Method: Fill each test tube about a quarter with water Take a small spoonful of each compound and put it in each test tube Give the test tubes a shake and record their solubility Results: SUBSTANCE PREDICTION SOLUBLE/INSOLUBLE Aluminium nitrate Soluble Soluble Aluminium sulphate Soluble Soluble Ammonium carbonate Insoluble Soluble? Ammonium chloride Soluble Soluble Ammonium sulphate Soluble Soluble Barium chloride Soluble Soluble Barium nitrate Soluble Soluble Seems soluble, still solid in the bottom, could have put too much in but apparently insoluble. Water is murky but insoluble Insoluble Barium sulphate Calcium carbonate Insoluble Calcium chloride Soluble Soluble Calcium hydroxide Insoluble Soluble? Calcium sulphate Insoluble Insoluble Cobalt chloride Soluble Soluble Copper II carbonate Insoluble Soluble? Copper II oxide Insoluble Copper II sulphate Soluble Soluble Iron II chloride Soluble Insoluble? Iron III chloride Soluble Soluble Iron III oxide Soluble Lead chloride Insoluble Insoluble Lead nitrate Soluble Soluble Lithium chloride SUBSTANCE Soluble Low solubility SOLUBLE/INSOLUBLE Magnesium chloride Soluble Soluble Magnesium hydroxide Insoluble Soluble? Magnesium nitrate Soluble Soluble PREDICTION Magnesium oxide Insoluble Magnesium sulphate Soluble Soluble Nickel sulphate Soluble Soluble Potassium carbonate Soluble Soluble Potassium chloride Soluble Soluble Potassium dichromate Soluble Soluble Potassium hydroxide Soluble Soluble Potassium iodide Soluble Soluble Potassium sulphate Soluble Soluble Silver nitrate Soluble Soluble Sodium carbonate Soluble Soluble Sodium chloride Soluble Soluble Sodium hydroxide Soluble Soluble Sodium nitrate Soluble Soluble Sodium sulphate Soluble Soluble Strontium chloride Soluble Soluble Discussion: 1. For the substances Barium sulphate, Calcium carbonate, Calcium sulphate and Lead chloride, it was predicted that they would be insoluble, which they were. The predictions were made based on the table of rules. For example, when sulphate is mixed with barium, it is one of the few times sulphate becomes insoluble. Iron II chloride on the other hand was predicted soluble, because according to the rules, chloride is soluble with any iron. But, in the prac it turned out to be insoluble. This could be because of reasons of inaccuracy such as putting too much of the substance in the test tube or not shaking the test tube enough, so the substance couldn’t all dissolve. 2. According to the table of rules, Sodium chloride is soluble. This diagram shows how sodium chloride dissolves in water. H+ O- Cl- H+ H+ H+ O- Cl- Cl- H+ H+ O- H+ Na+ Na+ Sodium, Na+, is a positive ion and Chlorine, Cl-, is a negative ion. The positive hydrogens of water, H+, attract and attach to the negative Chlorine. The same happens with Sodium and the oxygen part of water. O - H+ Na+ Hydrogen shells are formed around the separated molecules and prevent them from rejoining and so can no longer form a solid. 3. The majority of our results matched up to our predictions. The ones that didn’t were: Ammonium carbonate, Calcium hydroxide, Copper II carbonate and Magnesium hydroxide. They were predicted insoluble, using the table of rules, but in the prac were soluble. This could once again be because of inaccuracy, possibly not assessed well enough but it is more difficult for something to dissolve in something than to not – saying that, if something’s soluble it’s usually soluble, it is hard to get that wrong, but if something seems insoluble, there’s usually something you can do to make the substance dissolve better. Conclusion: We have now learnt to make accurate predictions of the solubility of a substance, using the table of solubility rules. Unpredicted outcomes have occurred and may not be because of wrong predictions. Inaccurate testing may have been the reason.