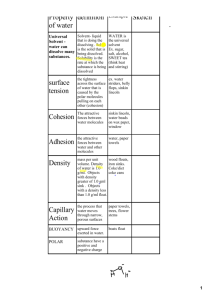
Properties of Water Review
advertisement

Analyze Water Properties. Name: 1. How does the colored water get into the leaves? 2. What properties of water allow for this to happen? 3. Why is this important for life? 4. Why doesn’t oil & water mix? 5. What property of water creates this scenario? 6. Why is this important for life? 7. What property allows water to bead up? 8. Why is this important for life? 9. Why can water dissolve so many particles? 10. What property allows water to do this? 11. Why is this important for life? 12. Water will bead up on wax paper but spread out on glass. Explain this. 13. Why does water resist vaporization? 14. Why is this important for life? 15. How does water react to changes in temperature? Does water heat up quickly or slowly? 16. Why is this important for life? 17. Draw 10 water molecules, properly oriented, hydrogen bonded to one another. 18. Some adult insects are unable to swim but are able to walk on top of water. What characteristic of water enables these insects to walk on top of water? A. pH C. atomic bonds B. solvent properties D. surface tension 19. Which term refers to water’s attraction to other substances that have full or partial electrical charges? A. polarity D. solvent B. adhesion E. density C. cohesion 20. Which term refers to water having partial positive charges and a partial negative charge? A. cohesion D. good solvent B. surface tension E. adhesion C. polarity 21. Which term refers to water’s attraction to different substances? A. polarity D. good solvent B. cohesion E. adhesion C. surface tension 22. Which term refers to the fact that water can dissolve polar molecules like sugar or ionic substances like salt? A. surface tension C. polarity B. high heat capacity D. cohesion 23. Which part(s) of a water molecule has/have a partial positive charge? A. the two hydrogen (H) atoms C. the one hydrogen (H) atom B. the two oxygen (O) atoms D. the one oxygen (O) atom 24. Which characteristic of water allows a paperclip to remain “floating” on water when the paperclip is gently placed on top of the water (even though paperclips are denser than water)? A. adhesion D. nonpolar B. surface tension E. inorganic C. solvent properties 25. Plants need to move water from their roots to their leaves in order to perform photosynthesis. The tube system that moves the water is called xylem. What property of water allows water to move up the xylem into the leaves? A. Evaporation C. Specific Heat B. Three phases of water, solid, liquid, or D. Capillary Action gas 26. Water has a high specific heat capacity; this means that it takes a lot of energy to change the temperature of water. Which of the following explains why this is important to life? a. Allow water to boil faster than other substance such as oil. b. Helps organisms to maintain a constant internal temperature. c. Helps organisms maintain an osmotic balance. d. Helps organisms transport substance throughout the organisms.