Notes: Water Chemistry
advertisement

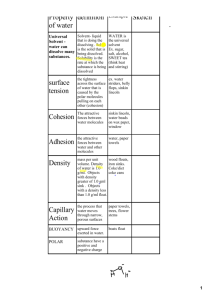
Notes: Water Chemistry Water • Water is the most important COMPOUND in living organisms! – Most organisms are made up of 70-95% of water • Water serves as a form of TRANSPORTATION – Helps move nutrients and other substances in and out of cells • Water is a POLAR molecule What is a Polar Molecule? • POLAR: A molecule with a POSITIVE end and a NEGATIVE end • Water molecules are attracted to each other by hydrogen bonds – Remember: van der Waals forces Water Structure Notice that opposite charges attract! Water Bonding • COHESION – an attraction between the SAME kind of substances – In water: a result of hydrogen bonds – Attraction between water molecules causes SURFACE TENSION Water Bonding • ADHESION – an attraction between DIFFERENT substances Capillary tubes Water & Glass Properties of Water 1. High surface TENSION 2. Creeps up in thin tubes - CAPILLARY action 3. High heat of VAPORIZATION 4. Resists TEMPERATURE change 5. Water EXPANDS when it freezes 6. Universal SOLVENT 1. High Surface Tension • Water is STICKY – Due to cohesion— sticks to itself • When hydrogen bonds are broken they quickly reform Water striders using water surface tension when mating. Surface Tension Causes the drop into a sphere 2. Capillary Action • MOVEMENT of water within the spaces of a porous material due to adhesion • Occurs because water is “STICKY” to another substance • Limited by gravity and the diameter of the capillary (tube) – Smaller the diameter, the greater the effect Capillary Action Capillary action causes the water to "climb" up this paper towel. Water Movement in Plants • Water moves upward through a plant from roots to leave through a COMBINATION of: – Cohesion – Capillary Action (adhesion) 3. High Heat of Vaporization • Water needs a LOT of heat to make liquid water change to a gas state (540 calories) 4. Resists Temperature Change • Water can ABSORB a tremendous amount of heat – Moderates our climate – It also takes a long time to cool down • In organisms, the ability to control temperature enables cells to maintain a constant internal temperature--Homeostatsis Water Stores Heat • Water heats more slowly and RETAINS heat LONGER than many substances • Many organisms release excess heat through evaporation – Sweating carries heat away from the body (HOMEOSTASIS) 5. Water Expands when it Freezes • Water EXPANDS when it freezes • Ice is LESS dense than liquid water • Ice FLOATS in liquid water 6. Universal Solvent • Many substances DISSOLVE in water – Commonly called the universal solvent. Why? • ....remember POLARITY of water – Because of this, water in nature and in use is rarely pure • Many substances are INSOLUBLE in water. Why? • They are NOT POLAR and not attracted to water