LAB TWO Enzyme Catalysis-2
advertisement

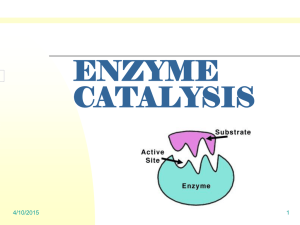
Catalase as a Model for Enzyme Catalysis and Reaction Rate Abstract: Introduction: Metabolism is the sum of chemical reactions occurring within an organism or a cell (Losos pg G-13). These reactions include a host of processes from simple reactions to complex biochemical pathways. Although these reactions can occur spontaneously, they occur too slowly for the products to be efficiently utilized by the cell. To survive, cells rely on enzymes to catalyze reactions. In essence, an enzyme is generally a protein that catalyzes or increases the rate of a chemical reaction. Enzymes bind to substrates at active sites. The individual amino acids of an enzyme result in specificity for one type of substrate. Shape of an enzyme also determines which substrates it is capable of bonding with. The fit is not perfect and small changes of the active site occur when it bonds to a substrate. This is known as induced fit. (Holtzclaw pg 3-5) Despite this change, enzymes do return to their original shape and are able to be reused over and over again. (Lab Manual pg 19) Enzymes will only function in optimal conditions. The salinity, pH, and temperature are several factors that can cause a protein to denature, or unfold, rendering it inactive. The presence of activators increases the rate of enzyme catalyzed reactions. Inhibitors do the opposite by slowing or halting a chemical reaction. Both activators and inhibitors allow the cell to control the rate at which chemical reactions occur. (Lab Manual pg 19-20) The initial rate of an enzyme catalyzed reaction should be linear when graphed. This is because with a large amount of substrate, there is a large amount of substrate to enzyme contact. The rate of a reaction is based on this initial rate and can be calculated by the equation y / x. This equation is the slope of the linear portion of the graph of time vs. products. (Lab Manual pg 19-21) Based on the above information we hypothesized the following; If the enzymes react with the substrate for a longer time, then more substrate will be used up because the substrates will be in contact with the enzymes longer thus creating more products. Purpose: The first purpose of this experiment is to calculate effects of changes in temperature, pH, enzyme and substrate concentration on enzyme catalyzed reaction rates and to understand how these changes affect the reaction rates. The second purpose is to calculate the rate of reaction of hydrogen peroxide with catalase. (Lab Manual pg 19) Results: Exercise 2B: The Base Line Assay Table I: Base Line Calculation Base Line Calculation Final Reading of Burette 37.1 mL Initial Reading of Burette 28.1 mL Base Line (Final –Initial) 9 mL KMnO4 Exercise 2C: The Uncatalyzed Rate of H2O2 Decomposition Table II: Uncatalyzed H2O2 Decomposition Uncatalyzed H2O2 Decomposition Final Reading of Burette 32 mL Initial Reading of Burette 22.9 ml Amount of H2O2 Spontaneously Decomposed (mL Baseline – mL KMnO4) -.1 ml Percent of H2O2 That Spontaneously Decomposed in 24 Hours [(mL Baseline –mL 24 hours)/mL Baseline] X 100 -1.11% Exercise 2D: An Enzyme-Catalyzed Rate of H2O2 Decomposition Table III: KMnO4 (ML) Base Line (mL) Final Reading (mL) 10 9 32.8 Time (Seconds) 30 60 9 9 33 33.4 90 9 120 9 180 9 360 9 33.5 33.8 44.8 45.2 Initial Reading (mL) Amount of KMnO4 Consumed Amount of H2O2 Used 32 32.8 33 33.4 33.5 44.2 44.8 0.8 0.2 0.4 0.1 0.3 0.6 0.4 8.2 8.8 8.6 8.9 8.7 8.4 8.6 Graph I: Product Formed Individual Data Product Formed (Individual Group Data) 12 R² = 0.1313 10 8.8 8.6 8.9 8.7 8.2 Product 8 8.6 8.4 Product Formed 6 Linear (Product Formed) 4 Linear (Product Formed) 2 0 0 0 100 200 Time (Seconds) 300 Graph II: Product Formed Class Data 400 Product Formed (Class Data) 12 Product Formed 10 9.4 8 9.3 9.7 9.4 R² = 0.8597 9.4 7.7 6 5.9 Series1 Poly. (Series1) 4 2 0 0 0 10 30 60 90 120 Time (Seconds) 180 360 References: Losos, Jonathan B. et al. 2008. Biology, 8th ed. New York, New York: The McGraw-Hill Companies, Inc Lab Manual - LAB TWO Enzyme Catalysis Holtzclaw, Theresa. "Enzyme Catalysis." The Biology Place. Person Education Inc, n.d. Web. 13 Oct. 2011. <http:/http://www.phschool.com/science/biology_place/ labbench/lab2/intro.html>.