KEY
advertisement

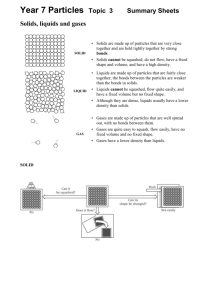
Unit 1 – Matter Some Basic Definitions: Qualitative non-numeric information often based on observation Quantitative numeric measurable information Observation qualitative information Interpretation an attempt to put meaning to an observation Description a list of properties of something Data quantitative information that is experimentally determined Experiment a test or procedure that is carried out to discover a result Hypothesis a single, unproven assumption which attempts to explain a natural phenomenon Theory is a set of hypothesis that ties together a large number of observations into a logical consistent and understandable pattern. It is a theory that has been TESTED and REPLICATED successfully many times Law is a broad generalization or summary statement which describes a large amount of experimental evidence stating how nature behaves wen a particular situation occurs. It has been “proven” Answer the following questions: 1. 2. 3. Give both a qualitative and a quantitative description of a) a length of time b) a temperature a) Soon, 5 min b) warm, 24C ( qualitative, quantitative) Copper is a reddish-colored element with a metallic lustre. It is an excellent conductor of heat and electricity, melts at 1085C and boils at 2563C. Archeological evidence shows that is has been minded for the past 5000 years and presently is considered to be one of the most important metals available. Copper is insoluble in water and virtually all other solvents, reacts easily with nitric acid but only slightly with sulphuric acids. Its density is 8.92g/mL, which makes is more dense that iron. What is the difference between a) observation and data, b) hypothesis and theory c) theory and law. A) observations are qualitative and data is quantitative b) confidence is low in an hypothesis but high in a theory. Hypothesis attempts to explain one experiment, theory results from may experiments c) theory is not or can not be proven, however a law can and has been. Laws describe what will happen in a known situation, but a theory attempts to explain a wide range of experimental results. The Physical Properties of Matter: Matter: anything that has mass and occupies space Chemistry: is the science concerned with the properties, composition, and behaviour of matter Substance: is something with a unique and identifiable set of properties (diff properties = diff substance) Physical Property: is a property of a substance that can be found without creating a new substance Ex. Density, color, hardness, mp, bp Chemical Property: is the ability of a substance to undergo chemical reactions and change into new substances either by itself or with other substances Ex. Hydrogen burns in air Intensive Property: physical property that depends solely on the nature of the substance NOT how much is present Ex. density, mp, bp, Extensive Property: physical property that depends on the amount of the substance present Ex. mass, *** are not used to identify the substance Answer the following questions: 1. 2. Which of the following describes physical or chemical properties a) glass is transparent physical b) salt melts at 801C physical c) adding lye to fat makes soap chemical d) copper conducts electricity physical e) fumes from ammonia and hydrochloric acid mix to produce a white smoke chemical Which are intensive or extensive properties: a)shape b)smell c)length d) color e)electric conductivity f)time required to dissolve a solid g) hardness intensive = b, d, e, g, and extensive = a, c, f Matter exists in three common states or “phases” each of which has a unique set of properties Phase or State Solids Properties Rigid Don’t readily change shape Very small changes to volume when heated or pressure = Not compressible Conform to the shape of their container Very small changes to volume when heated or pressure =Not compressible Liquids Gases Conform to the shape of their containers Compressible – drastic changes in volume when heated or pressure Molecular Dispersement Particles are packed into a given volume in a highly organized rigid manner which requires the particles to be in direct contact with one another. Particles are in close contact with one another but have enough room to slide past one another easily and prevent an organized packing The particles are widely separated and only contact each other in collisions Liquids are an “intermediate” phase between solids and gases because: Like a gas because conform to space of container Like a solid because not compressible Other Definitions you need to know: Hardness: ability of a solid to reseit abrasion or scratching Malleability: : ability to be rolled or hammered into thin sheets Ductility: : ability to be stretched or drawn into wires Lustre: : the manner in which a solid surface reflects light - metallic, glassy, oily, pearly, silky, dull Viscosity: : the resistance to a fluid flow Diffusion: : the intermingling of fluids as a result of motion within the fluid (liquids and gases Vapour: : the gaseous material formed by the evaporation of a substance which boils above room temperature Boiling Point: : the temperature at which a liquid changes to a solid Melting Point: : the temperature at which a solid changes to a liquid Answer the following Questions: 1. 2. 3. Compare the relative boiling points of ionic and covalent compounds? Ionic compounds havehigh boiling points compared to covalent compounds What are intermolecular and intramolecular bonds? An intramolecular force is any force that holds together the atoms making up a molecule or compound. They contain all types of chemical bond. They are stronger than intermolecular forces, which are present between atoms or molecules that are not actually bonded. Explain your answer to question 1, using the terms defined in question 2. – The boiling point is essentially the temperature at which there is enough kinetic molecular energy to separate the molecules farther from one another so that they are in a gas state. This temperature essentially must overcome the intermolecular forces between the molecules which are stronger between ionic compounds that covalent compounds. Recall that ionic compounds contain positive and negative ions. This results in them having not only intra but also intermolecular attractions which must be overcome with kinetic energy in order to have them spread apart (change phase). Classification of Matter: System:_is the part of the universe being studies in a given situation Phase:___is any part of a system which is unitform in both its compositions and properties Element: is a substance which cannot be separated into simpler substances as a result of any chemical process Ex. silver metal, Copper metal, hydrogen gas Atom is the smallest possible unit of an element which retains the fundamental properties of the element Ex. silver (Ag) Copper (Cu) Hydrogen (H) Molecule is a cluster of two or more atoms held together strongly by electrical forces Ex. water, ethanol, table sugar Ion an atome or molecule which possesses an electric charge Ex. sodium ion, nitrate ion Particle is a general erm used to describle a small bit of matter such as an atome, molecule or ion Homogeneous has uniform unchanging physical properties Ex. 1L or an aqueous solutions which has a consistant clarity and density of 1.035g/mL throughout. Heterogeneous the substances making up the mixture will each have their own, unique set of properties Ex. water and iron mixture, both maintain their unique density and boiling points Pure substance unchanging uniform physical properties Ex. mercury in a thermometer Mixture a system made up to two or more substances such that the relative amounts of each can be varied Ex. salt dissolved in water Mechanical mixture is a heterogeneous mixture Ex. gravel, a pencil Solution a homogeneous mixture of two or more substances Ex. air, soda pop, alloys Solvent component in a solution which exists in the greater quanitity Ex. salt dissolved in water water is the solvent Solute is the component in a solution which exsists in the smaller quantity Ex. the salt Compound is a pure substance make up of two or more types of atoms but there is only one type of molecule in a compound Ex. sugar, salt, water Separation of Substances: Miscible: two liquids that are mutually soluble in each other in all proportions Immiscible: two liquids that are insoluble in one another Synopsys of Separations Methods – (refer to last day’s notes) Mechanical Mixtures: MIXTURE Solid in Solid METHOD Hand Separation Gravity Separation Solvent Extraction WHEN TO USE THE METHOD Large chunks present among other solids The density of desired solid is much diff from the other solid One solid preferentially dissolves in a particular solvent Chromotography Solid in Liquid Hand Separation Gravity Separation Filtration The solids are colored, present in small amounts and soluble in some solvent A few large pieces of solid are present in the liquid Solid particles are preens tin small amount of liquid Solid particles are present in a large amount of liquid Solutions: MIXTURE Solid in a Liquid METHOD Evaporation Distillation Solvent Extraction Solid in a liquid Con’t Recrystallization Chromotography Liquid in Liquid Distillation Solvent Extraction WHEN TO USE THE METHOD The solid is wanted and the liquid is not The liquid is wanted, the solid may not be An immiscible added solvent preferentially dissolves at least one but not all solids One dissolve solid is much less soluble that the others (liq not wanter) Small amounts of more that one colored solid are present (liq not want) Two or more liquids are present and have different boiling temperatures In immiscible added solvent preferentially dissolves at least one but not all of the liquids present Phase Changes: Watch video: http://www.youtube.com/watch?v=KCL8zqjXbME Chemical Change: is a change in which a new substance is formed with a new set of properties Physical Change: is a change in the phase of a substance such that no new substance is formed ** Chemical changes are frequently accompanied by physical changes Melting temperature solid to liquid Freezing temperature liquid to solid Boiling Temperature liquid to gas Condensation gas to liquid On the sloping portions of the graph: all the heat is used to warm the substance so the temperature rises the heat is going directly into increasing the kinetic molecular energy of the compound and thus the temp increases On the level portions of the graph: the heat added is used to break up the solid and allow the liquid to form. A better way to say this is that the energy is being used to break the attraction between the molecules rather that adding kinetic energy to them which we see as an increase in temperature Sublimation: solid to gas Deposition: gas to solid Kinetic Energy’s Role in Physical Change: Watch: http://education-portal.com/academy/lesson/changes-in-heat-and-energy-diagrams.html#lesson Temperature Energy Kinetic Energy – the energy molecules possess as a result of their motion Heat Heat is the total energy of molecular motion in a substance while temperature is a measure of the average energy of molecular motion in a substance. Heat energy depends on the speed of the particles, the number of particles (the size or mass), and the type of particles in an object. Temperature does not depend on the size or type of object. For example, the temperature of a small cup of water might be the same as the temperature of a large tub of water, but the tub of water has more heat because it has more water and thus more total thermal energy.