Scientific Report
advertisement

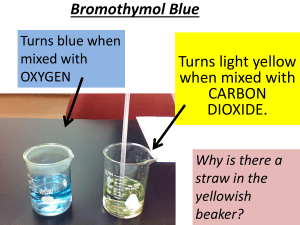
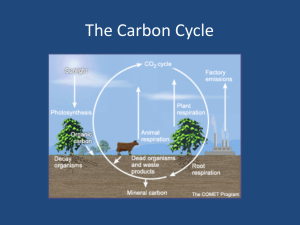
Ren Lim Tutor : Abofazyl Bayat The Carbon Cycle & Global Warming Carbon Carbon is the 6th most abundant element in the universe [1]. Its presence tells us that compound is organic while its absence lets us know that that compound is inorganic. Every living organism in this planet either has carbon atoms for structure and support or utilizes carbon as a source of energy. Carbon can be found in the form of CaCO3 (limestone), graphite or diamond and exists in the Earth's atmosphere primarily as the gas carbon dioxide (CO2). Although carbon is found a small percentage of the atmosphere (approximately 0.04% on a molar basis), it still plays a vital role in supporting life. Our forests store 86% of the planet's above-ground carbon and 73% of the planet's soil carbon.[2] The Cycle Figure 1. A cartoon of the global carbon cycle. Pools (in black) are gigatons (1Gt = 1x109 Tons) of carbon, and fluxes (in purple) are Gt carbon per year. Illustration courtesy NASA Earth Science Enterprise. The movement of carbon in its many forms in between oceans, seas, rivers, the biosphere and etc. is known as the carbon cycle. The global carbon cycle can be divided into two categories: the geological, which operates over large time scales (millions of years), and the biological/physical, which operates at shorter time scales (days to thousands of years). The geographical part Billions of years ago, as planetesimals (small bodies that formed from the solar nebula) and carbon-containing meteorites bombarded our planet’s surface, the carbon content of the solid Earth steadily increased. Since those times, carbonic acid (a weak acid derived from the reaction between atmospheric carbon dioxide and water) has slowly but continuously combined in the Earth’s crust to form insoluble carbonates (carbon-containing compounds with other minerals in it) through a process called weathering. [4]. Then through erosion, these carbonates are carried in surface waters like streams and rivers eventually to the ocean, where they precipitate out to form minerals like calcite (CaCO3). Through continued deposition and burial, this calcite sediment forms a type of rock called limestone. The cycle continues as seafloor spreading pushes the seafloor in a process called subduction. In this process, carbon is pushed deeper into the earth by tectonic forces, causing it to heat up and melt. And when this carbon rises back up to the surface, it releases CO2 and returns to the atmosphere. [3] The chemical part Biology also plays an important role in the carbon cycle through the process of respiration and photosynthesis. In fact, the amount of carbon taken up by photosynthesis and released back to the atmosphere by respiration each year is 1,000 times greater than the amount of carbon that moves through the geological cycle on an annual basis. [4] Figure 2- How plants takes in CO2 as a for photosynthesis (Illustration courtesy http://earthobservatory.nasa.gov/Features/CarbonCycle/ carbon_cycle2.php - P.J. Sellers et al.) During photosynthesis, plants absorb carbon dioxide and sunlight to create fuel—glucose and other sugars—for building plant structures. This process forms the foundation of the biological carbon cycle as shown in figure 2. Plants and animals in turn use these carbohydrates through respiration as a fuel for energy. Respiration and decomposition then returns fixed carbon back to the atmosphere. Photosynthesis and respiration also play important role in the long-term geological cycling of carbon. The presence of land vegetation enhances the weathering of soil, leading to the long term but slow uptake of CO2 in atmosphere. [3] Respiration: C6H12O6 (organic matter) + 6O2 6CO2 + 6 H2O + energy - equation [1] Photosynthesis: energy (sunlight) + 6CO2 + H2O C6H12O6 + 6O2 - equation [2] Equation 1 & 2 – The equation for both respiration and photosynthesis which accounts for how most of the carbon in the carbon cycle is being transported around the biogeochemical cycle. In oceans, some of the carbon is taken up by microscopic plants called phytoplankton to make shells which are composed of calcium carbonate. These shells settle to the bottom when these organisms die to form sediments. When photosynthesis exceeds respiration, organic matter build up over millions of years to form coal and oil deposits. This reduces the content of carbon dioxide from the atmosphere over time. [3] Since the industrial revolution 150 years ago, with the increased burning of fossil fuels and the effects of deforestation there was a long term rise in atmospheric CO2. This intake of CO2 by plants cannot keep in pace with CO2 output which is being released by the burning of fossil fuels by humans thus leading to a CO2 net increase. As such the CO2 concentrations in the atmosphere are higher today than how they have been over the last half-million years. [5] Recently, scientists have studied both short- and long-term measurements of atmospheric CO2 levels. Charles Keeling, is responsible for creating the longest continuous record of atmospheric CO2 concentrations, taken at the Mauna Loa observatory in Hawaii. His data which is now widely known as the “Keeling curve,” showed that human activities are significantly altering the natural carbon cycle. [6] CO2 is a greenhouse gas and scientists believe that the increase in CO2 is already causing important changes in the global climate. The observed 0.6 degree C increase in global average temperature over the past century has been attributed to the increases in atmospheric CO2. It is also estimated that there will be a mean global temperature increase between 1.4 and 5.8 degrees C over the next century as a result of the increase CO2 and greenhouse gases further increase. This would cause significant rise in average sea-level (0.09-0.88 meters), exposing low-lying coastal cities or cities located by to increasingly frequent and severe floods. Glacial retreat and species range shifts are also likely to result from global warming, and it would also be interesting to see whether trees can shift quickly enough in order to cope with climate change. [7] Even without the temperature increase, the increase in concentrations of CO2 can change the growth patterns of plants worldwide. This is because some species of plants respond to CO2 increment better than others. an important impact on patterns of plant growth worldwide. So without a change in temperature, the flora of the world will change [8] Understanding and mitigating the negative impact of atmospheric CO 2 constitute two of the most central challenges that environmental scientists and policy makers currently face [9] .In order to address this issue, the scientific community has formed the Intergovernmental Panel on Climate Change (IPCC), orating to produce consensus reports on climate change science. [10] Also, many nations have agreed on the terms of the Kyoto Protocol, a treaty which is aimed at averting human-induced carbon dioxide emission. But problems arises still such as the fact that although United States is responsible for approximately one quarter of global CO2 emissions , it still declined to participate in the Kyoto Protocol. References [1] -http://education.jlab.org/itselemental/ele006.html, on carbon being the 6th most abundant element in the universe [2] - Sedjo, Roger.1993. The Carbon Cycle and Global Forest Ecosystem. Water, Air, and Soil Pollution 70, 295-307. (via Oregon Wild Report on Forests, Carbon, and Global Warming), on the composition of carbon outside the athmosphere [3]-http://66.102.9.132/search?q=cache:PCy4sFs868J:151.200.174.40/flinthill/homework/ShoffTai/documents/CarbonCycle.ppt+amount +of+carbon+dioxide+released+by+respiration+1000+times&cd=15&hl=en&ct=clnk&gl=uk &client=firefox-a on how much more carbon dioxide is being released by respiration compared to being transported around [4] -http://earthobservatory.nasa.gov/Features/CarbonCycle/. The main article for the Geological and biological processes for carbon cycle. [5] - http://news.mongabay.com/2005/1124-climate.html on the levels of carbon dioxide today being higher than it has always been for 650000 years. [6] - http://www.aip.org/history/climate/co2.htm, on the discovery of global warming by Keeling with his data included as well [7] -http://timeforchange.org/co2-concentration-causing-temperature-increase, on carbon dioxide as a greenhouse gas and its increase in its concentration on the temperature increase of the atmosphere [8] -http://www.futurepundit.com/archives/001938.html, basis of information on how certain plants flourishes in the presence of CO2 [9] http://www.nature.com.libproxy.ucl.ac.uk/scientificamerican/journal/v302/n4/full/scien tificamerican0410-58.html on challenges of policy making to combat climate change [10] - Further reading on how to solve climate change can be found in reference http://www.ipcc.ch/organization/organization.htm, IPCC Note :- Excluding references, this is a 1140 words essay.